Introduction to Atyr Pharma Inc. (ATYR) Securities Class Action Lawsuit
A federal securities class action has been filed against Atyr Pharma Inc. (NASDAQ: ATYR) covering January 16, 2025 to September 12, 2025, inclusive. Investors allege the company and its CEO misrepresented the efficacy of Efzofitimod and the Phase 3 EFZO-FIT study, especially around steroid reduction and the prospect of eliminating steroids. The complaint says defendants paired upbeat messaging with omissions about study design and factors that could enable steroid withdrawal independent of treatment. On September 15, 2025, Atyr disclosed that EFZO-FIT failed to meet its primary endpoint of change from baseline in mean daily oral corticosteroid dose at week 48. Following the disclosure, investors who bought during the class period allegedly suffered significant losses.
Atyr Pharma Inc. (ATYR) Securities Lawsuit Case Details
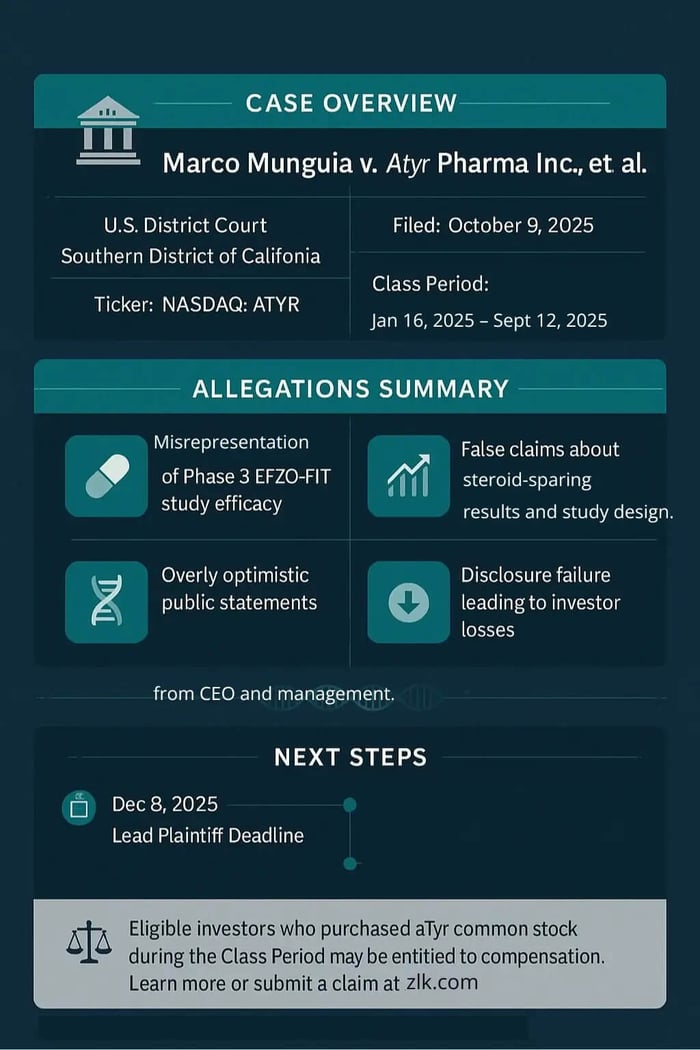
Case Name: Munguia v. Atyr Pharma Inc., et al.
Case No.: 3:25-cv-02681-WQH-SBC
Jurisdiction: U.S. District Court, Southern District of California
Filed on: October 9, 2025
Atyr Pharma Inc. (ATYR) Company Profile
Atyr is a clinical stage biotechnology company developing therapies for fibrosis and inflammation by translating tRNA synthetase biology. The company focuses on interstitial lung disease, leveraging a proprietary library of domains derived from all 20 tRNA synthetases.
Atyr Pharma Inc. (ATYR) Securities Lawsuit Class Period
January 16, 2025–September 12, 2025, inclusive.
Eligible investors include all who purchased or otherwise acquired Atyr common stock during the Class Period and may be eligible to join the Atyr Pharma Inc. (ATYR) class action lawsuit.
Allegations in the Atyr Pharma Inc. (ATYR) Securities Class Action Lawsuit
According to the complaint, Atyr Pharma Inc. and its President, Chief Executive Officer, and Director, Sanjay S. Shukla, targeted investors with a steady stream of optimism about efzofitimod and the Phase 3 EFZO-FIT study for pulmonary sarcoidosis. The lawsuit alleges they told a compelling story about a paradigm-shifting therapy and a Phase 3 catalyst while concealing adverse facts about the study's design and efficacy related to steroid tapering.
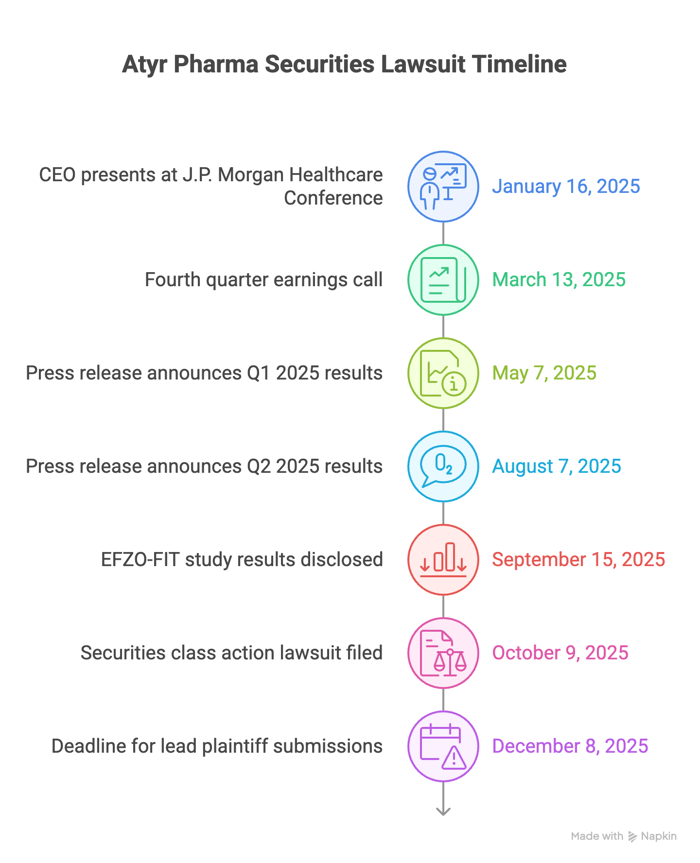
The narrative began on January 16, 2025, when Shukla presented at the 43rd Annual J.P. Morgan Healthcare Conference. He described efzofitimod as a "first-in-class biologic" in Phase 3 that was "generating fantastic results thus far," said the company had seen "dose response…in all of those end points we measured," and claimed they were seeing "quite remarkable steroid-sparing effects" in blinded reviews, even raising the prospect of reducing or "maybe even eliminate" steroids. He framed the program as a "real major Phase III catalyst" expected later that year.
The complaint alleges that defendants continued to provide overwhelmingly positive statements to investors during this period. On March 13, 2025, during the fourth quarter earnings call, Shukla stated, "We remain confident in the favorable safety profile we have seen for efzofitimod to date, which we believe is the key value proposition of the drug." On May 7, 2025, the Company issued a press release announcing Q1 2025 financial results and provided a corporate update that the Phase 3 EFZO-FIT study was on track to announce topline data in the third quarter. As part of that press release, aTyr stated that “[t]he primary endpoint of the study is steroid reduction measured as the absolute change from baseline to week 48.” Then on August 7, 2025, the Company issued a press release announcing Q2 2025 financial results and reiterated that topline data from the Phase 3 EFZO-FIT study was to be expected by mid-September 2025.
Meanwhile, investors allege that behind these statements, defendants concealed adverse facts about the EFZO-FIT study design and efficacy, gave the false impression that EFZO-FIT would meet its primary endpoint, and crafted a narrative that the study would enable patients to fully remove steroids. The complaint further alleges defendants failed to disclose that other factors could allow patients to eliminate steroids, which bore directly on the primary endpoint—change from baseline in mean daily oral corticosteroid dose at week 48.
The Truth Emerges
The story turned on September 15, 2025, when, pre-market, Atyr hosted an investor event and disclosed that EFZO-FIT "did not meet the primary endpoint of change from baseline in mean daily oral corticosteroid or OCS dose at week 48." Management acknowledged a "higher-than-anticipated placebo response" and said the company would engage with the FDA to determine a path forward given the disappointing topline results.
These admissions contradicted months of confident messaging about dose response across endpoints, "remarkable steroid-sparing effects," and a paradigm-shifting therapy. The disclosure marked a sharp break from the earlier narrative and realigned investor expectations with the study's topline outcome.
Market Reaction
As the disclosure hit on September 15, 2025, the market reacted immediately. Atyr's stock fell 83.2% in a single day—from a prior close of $6.03 per share on September 12, 2025 to $1.02 per share—an intraday decline of $5.01 per share. The announcement was made pre-market, and the move reflected the market's reassessment of EFZO-FIT's prospects after the failed primary endpoint.
Next Steps
- Submissions for lead plaintiff are due December 8, 2025.
- The Court will issue its order for lead plaintiff and counsel in the weeks after submissions are due.
- The Court will then consider motion for class certification.
- The Court will later consider a Motion to Dismiss.
To learn if you are eligible for recovery under the ATYR securities class action lawsuit, visit the case submission page here.
Disclaimer: This shareholder alert is for informational purposes only and does not constitute legal advice. Consult a qualified attorney for personalized guidance. No specific outcomes are guaranteed.
![Atyr Pharma Inc. (ATYR) Securities Class Action Lawsuit [October 23, 2025]](https://dropinblog.net/cdn-cgi/image/fit=scale-down,format=auto,width=700/34260815/files/featured/atyr-banner-image.webp)

![WPP PLC (WPP) Securities Class Action Lawsuit Filed [October 23, 2025]](https://dropinblog.net/cdn-cgi/image/fit=scale-down,format=auto,width=700/34260815/files/featured/wpp-banner-image.webp)
![Baxter International, Inc. (BAX) Securities Class Action Lawsuit Filed [October 23, 2025]](https://dropinblog.net/cdn-cgi/image/fit=scale-down,format=auto,width=700/34260815/files/featured/bax-banner-image.webp)
![Moonlake Immunotherapeutics (MLTX) Securities Class Action Lawsuit Filed [October 28, 2025]](https://dropinblog.net/cdn-cgi/image/fit=scale-down,format=auto,width=700/34260815/files/featured/mltx-banner-image-2.png)