Introduction to Cytokinetics, Incorporated (CYTK) Securities Class Action Lawsuit
A federal securities fraud class action has been filed against Cytokinetics, Incorporated (NASDAQ: CYTK) and its CEO, Robert I. Blum. The class period runs from December 27, 2023 to May 6, 2025, inclusive. Investors allege Defendants misrepresented the regulatory timeline for aficamten by signaling FDA approval in the second half of 2025, anchored to a September 26, 2025 PDUFA date, while concealing material risks tied to the absence of a required REMS in the original NDA. The truth surfaced when Cytokinetics disclosed an FDA-driven extension of the PDUFA action date to December 26, 2025 to review a REMS submitted after the initial filing, and management admitted they had filed the NDA without a REMS despite multiple FDA meetings discussing safety and risk mitigation. As the disclosures unfolded, the stock fell sharply, inflicting losses on investors who purchased at allegedly inflated prices.
Cytokinetics, Incorporated (CYTK) Securities Lawsuit Case Details
Case Name: Seidman v. Cytokinetics, Incorporated, et al.
Case No.: 3:25-cv-07923
Jurisdiction: U.S. District Court, Northern District of California
Filed on: September 17, 2025
Cytokinetics, Incorporated (CYTK) Company Profile
Cytokinetics is a biopharmaceutical company focused on discovering, developing, and commercializing first-in-class muscle activators and next-in-class muscle inhibitors for serious cardiovascular and neuromuscular diseases. Its lead program includes aficamten, a cardiac myosin inhibitor for obstructive hypertrophic cardiomyopathy within a specialty cardiology franchise.
Cytokinetics, Incorporated (CYTK) Securities Lawsuit Class Period
December 27, 2023–May 6, 2025, inclusive.
All investors who purchased or otherwise acquired Cytokinetics common stock during the Class Period are included in the putative class and may be eligible to join the Cytokinetics, Incorporated (CYTK) class action lawsuit.
Allegations in the Cytokinetics, Incorporated (CYTK) Securities Class Action Lawsuit
The lawsuit targets Cytokinetics, Incorporated and its Chief Executive Officer, President, and Director, Robert I. Blum. According to the complaint, Defendants told investors they were on track for aficamten's NDA review and approval in the second half of 2025, tied to a September 26, 2025 PDUFA date. Investors allege these statements painted a clear path to approval and a launch-ready posture while omitting material regulatory risks.
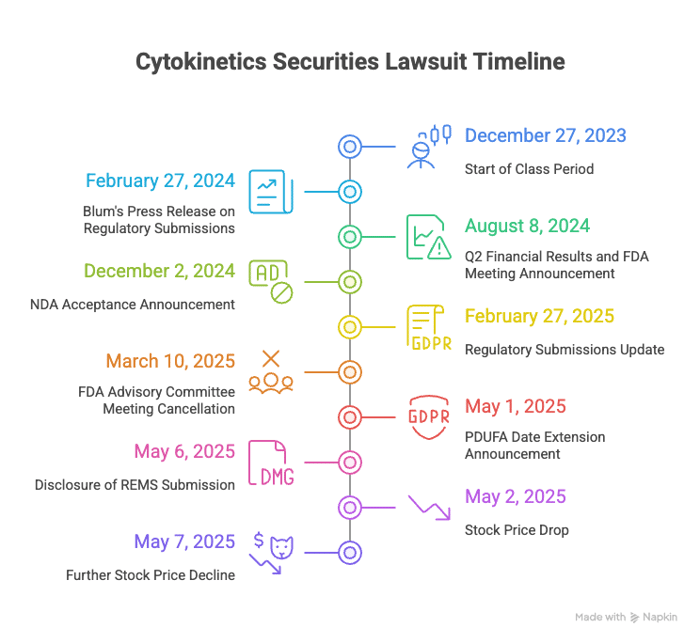
The narrative began on February 27, 2024, when Blum told investors in a press release that the company ended 2023 strong, was preparing regulatory submissions for aficamten, and was executing commercial readiness activities. On August 8, 2024, the company issued a press release announcing Q2 2024 financial results and said it had participated in a Type B FDA meeting about safety monitoring and risk mitigation and that a rolling NDA submission was underway, adding it expected to propose a "distinct risk mitigation approach" specific to aficamten.
After FDA accepted the NDA, Blum called the December 2, 2024 acceptance a “significant milestone.” Then, on February 27, 2025, he reiterated that regulatory submissions were on file in the U.S., Europe, and China, that regulatory reviews were underway, and that commercial readiness activities were on track to support planned launch activities.
Behind these messages, investors allege Defendants failed to disclose that the company had not included a REMS with the initial NDA despite multiple FDA interactions discussing safety and risk mitigation. The complaint alleges Defendants concealed the FDA's potential requirement for a REMS due to aficamten's intrinsic properties, which put the touted timeline at risk.
The Truth Emerges
The sequence began on March 10, 2025, when Cytokinetics filed a Form 8-K stating the FDA would not convene an advisory committee meeting to review the NDA for aficamten. Then, on May 1, 2025, the company announced after market hours that the FDA had extended the PDUFA action date from September 26, 2025 to December 26, 2025 to review a REMS that Cytokinetics submitted at the FDA's request after the initial NDA filing. Finally, on May 6, 2025, during an earnings call, management disclosed they had three meetings with the FDA before filing and nevertheless chose to submit the NDA without a REMS, acknowledging they understood REMS would be a focus of the agency's review.
These revelations cut against earlier assurances about regulatory progress and a second-half 2025 approval trajectory. The extension tied to a post-filing REMS submission contradicted prior statements about risk mitigation and underscored that the NDA had been filed without an accompanying REMS despite prior FDA discussions on safety and risk.
Market Reaction
The May 1, 2025 PDUFA extension announcement arrived after market close, and the stock fell the next trading day. On May 2, 2025, Cytokinetics' common stock dropped $5.57 per share, approximately 13%, to close at $37.35. After management provided additional details on May 6, 2025 about the FDA meetings and the decision to file without a REMS, the stock fell another $0.93 on May 7, 2025, closing at $33.04.
Next Steps
- Submissions for lead plaintiff are due November 17, 2025.
- The Court will issue its order for lead plaintiff and counsel in the weeks after submissions are due.
- The Court will then consider motion for class certification.
- The Court will later consider a Motion to Dismiss.
To learn if you are eligible for recovery under the CYTK securities class action lawsuit, visit the case submission page here.
Disclaimer: This shareholder alert is for informational purposes only and does not constitute legal advice. Consult a qualified attorney for personalized guidance. No specific outcomes are guaranteed.
![Cytokinetics, Incorporated (CYTK) Securities Class Action Lawsuit Filed [October 13, 2025]](https://dropinblog.net/cdn-cgi/image/fit=scale-down,format=auto,width=700/34260815/files/featured/cytokinetics-cytk-securities-lawsuit-blog-banner.webp)


![Fluor Corporation (FLR) Securities Class Action Lawsuit Filed [October 14, 2025]](https://dropinblog.net/cdn-cgi/image/fit=scale-down,format=auto,width=700/34260815/files/featured/flour-corporation-securities-lawsuit-blog-banner.webp)
![RCI Hospitality Holdings, Inc. (RICK) Securities Class Action Lawsuit Filed [October 16, 2025]](https://dropinblog.net/cdn-cgi/image/fit=scale-down,format=auto,width=700/34260815/files/featured/rci-hospitality-holdings-rick-securities-lawsuit-blog-banner.webp)
![KBR, Inc. (KBR) Securities Class Action Lawsuit Filed [October 16, 2025]](https://dropinblog.net/cdn-cgi/image/fit=scale-down,format=auto,width=700/34260815/files/featured/kbr-banner-image.webp)