Introduction to Dexcom, Inc. (DXCM) Securities Class Action Lawsuit
A securities fraud class action has been filed against Dexcom, Inc. (NASDAQ: DXCM) covering July 26, 2024 through September 17, 2025. Investors allege the Company and senior executives misrepresented the accuracy, reliability, and regulatory status of Dexcom's G6 and G7 continuous glucose monitoring (“CGM”) devices. According to the complaint, while executives touted improvements and "the most accurate CGM" claims, the company had made unauthorized design changes that allegedly rendered the devices less reliable and posed health risks. The truth surfaced through an FDA Warning Letter disclosed on March 7, 2025, and published on March 25, 2025, followed by September 2025 reports highlighting performance concerns and patient harm. As these facts emerged, Dexcom's stock declined on multiple dates, causing investor losses.
Dexcom, Inc. (DXCM) Securities Lawsuit Case Details
Case Name: Prime v. Dexcom, Inc., et al.
Case No.: 1:25-cv-08912
Jurisdiction: U.S. District Court, Southern District of New York
Filed on: October 27, 2025
Dexcom, Inc. (DXCM) Company Profile
Dexcom is a medical device company focused on the design, development, and commercialization of continuous glucose monitoring systems for diabetes and metabolic health. Its products include the Dexcom G6 and G7 CGM systems, with G7 serving as the flagship product.
Dexcom, Inc. (DXCM) Securities Lawsuit Class Period
July 26, 2024–September 17, 2025, inclusive.
All persons and entities other than Defendants that purchased or otherwise acquired Dexcom securities during the Class Period may be eligible to join the Dexcom, Inc. (DXCM) class action lawsuit.
Allegations in the Dexcom, Inc. (DXCM) Securities Class Action Lawsuit
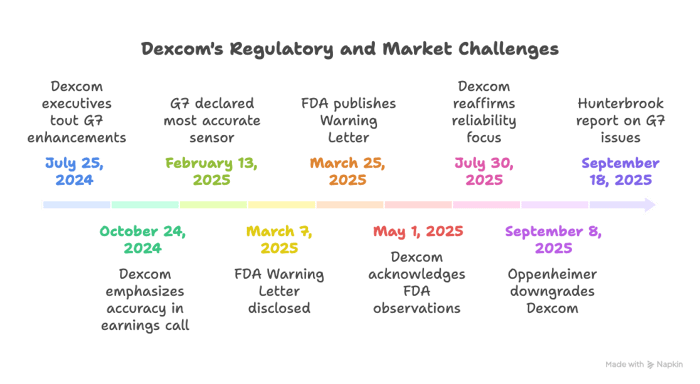
The complaint targets Dexcom, Inc. and senior executives Kevin R. Sayer, Jacob S. Leach, and Jereme M. Sylvain. Investors allege that throughout the period, Defendants overstated enhancements to G7 and the devices' accuracy, reliability, and functionality, while downplaying risks tied to design changes and manufacturing. On July 25, 2024, during a conference call, Sayer told investors "We've built upon the performance of G7, making it even better," citing software updates, a stronger adhesive, and expanded Bluetooth range, while that same day Sylvain highlighted migration to G7 and "steady progress" at high‑volume facilities in Mesa and Malaysia. Also on July 25, 2024, Dexcom filed its Form 10‑Q emphasizing product development focused on improved performance and intelligent insulin administration. On October 24, 2024, Sayer stated on an earnings call that "The accuracy of Dexcom is tried and true and proven to these patients," and that same day the company filed another Form 10‑Q repeating its development focus.
As the year turned, Dexcom issued a January 13, 2025 press release touting "key strategic investments" that "steadily progressed" and positioned the company for growth. On February 13, 2025, Leach told investors on the earnings call, "G7 is the most accurate sensor available," while acknowledging continued efforts to enhance accuracy and reliability. Days later, on February 18, 2025, the annual Form 10‑K described G7 as "the most accurate CGM cleared by the FDA" and highlighted alert features intended to predict and warn of dangerous glucose levels.
The narrative shifted in March. On March 7, 2025, Dexcom filed a Form 8‑K disclosing receipt of an FDA Warning Letter on March 4, 2025 following inspections in San Diego and Mesa. On March 25, 2025, the FDA published the Warning Letter stating the G6 and G7 CGM systems were "adulterated" because Dexcom lacked approved premarket applications for the modifications. On May 1, 2025, Sayer told investors the company took the FDA's observations seriously and had begun corrective actions, and that same day Dexcom filed its Form 10‑Q reiterating its product development focus. On July 30, 2025, Leach said on the Q2 2025 call that Dexcom needed to keep building sensors that are "reliable" and "accurate," and the company filed its July 30, 2025 Form 10‑Q. By September 8, 2025, Oppenheimer issued a downgrade, citing field checks flagging rising concerns about G7 accuracy and performance; and on September 18, 2025, a Hunterbrook report described severe G7 device issues, including hospitalizations and deaths linked to inaccurate readings.
According to investors, the reality behind these statements was a series of unauthorized FDA design changes to the G6 and G7 that allegedly reduced reliability and presented material health risks. The complaint alleges Defendants overstated enhancements and accuracy, downplayed scope and severity, and exposed the company to heightened regulatory scrutiny, enforcement, and harm.
The Truth Emerges
The sequence of corrective disclosures began on March 7, 2025, when Dexcom filed a Form 8‑K revealing it had received an FDA Warning Letter on March 4 following facility inspections. The picture sharpened on March 25, 2025, when the FDA published the letter stating the G6 and G7 systems were adulterated due to unapproved modifications, contradicting earlier assurances of accuracy and improvements. On May 1, 2025, Sayer acknowledged the FDA's observations on the earnings call and said the team had begun corrective actions.
In early September 2025, third-party reports and analysis added further detail. On September 8, 2025, Oppenheimer downgraded Dexcom after field checks pointed to rising concerns about G7 accuracy, failed insertions, abrupt stoppages, and other issues. On September 18, 2025, Hunterbrook published "Dexcom's Fatal Flaws," reporting that G7 users had been hospitalized and died following inaccurate readings, directly clashing with prior characterizations of G7 as the "most accurate" and reliable system.
Market Reaction
The market responded in steps as these disclosures landed. After Dexcom disclosed the FDA Warning Letter in a March 7, 2025 SEC filing, the stock fell $7.12 per share, or 9.15%, to close at $70.72 on March 10, 2025. When the FDA published the Warning Letter on March 25, 2025, Dexcom's stock fell $3.19 per share, or 4.24%, closing at $72.13 on March 26, 2025. On September 8, 2025, following Oppenheimer's downgrade, the stock declined $2.51 per share, or 3.12%, to close at $78.00. And as the September 18, 2025 Hunterbrook report circulated, the stock fell $8.99 per share, or 11.76%, to close at $67.45 on September 19, 2025.
Next Steps
- Submissions for lead plaintiff are due December 26, 2025.
- The Court will issue its order for lead plaintiff and counsel in the weeks after submissions are due.
- The Court will then consider motion for class certification.
- The Court will later consider a Motion to Dismiss.
To learn if you are eligible for recovery under the DXCM securities class action lawsuit, visit the case submission page here.
Disclaimer: This shareholder alert is for informational purposes only and does not constitute legal advice. Consult a qualified attorney for personalized guidance. No specific outcomes are guaranteed.
![Dexcom, Inc. (DXCM) Securities Class Action Lawsuit Filed [November 5, 2025]](https://dropinblog.net/cdn-cgi/image/fit=scale-down,format=auto,width=700/34260815/files/featured/dxcm-banner-image-v2.png)



![James Hardie Industries plc. (JHX) Securities Class Action Lawsuit Filed [November 5, 2025]](https://dropinblog.net/cdn-cgi/image/fit=scale-down,format=auto,width=700/34260815/files/featured/jhx-banner-image.png)
![Avantor, Inc. (AVTR) Securities Class Action Lawsuit Filed [November 5, 2025]](https://dropinblog.net/cdn-cgi/image/fit=scale-down,format=auto,width=700/34260815/files/featured/avtr-banner-image-2.png)
![Marex Group plc (MRX) Securities Class Action Lawsuit Filed [November 6, 2025]](https://dropinblog.net/cdn-cgi/image/fit=scale-down,format=auto,width=700/34260815/files/featured/mrx-banner-image-2.png)