Introduction to Sana Biotechnology, Inc Lawsuit
This is a shareholder alert about the enCore Energy Corp. class action lawsuit filed on or about March 21, 2025. The lawsuit seeks to recover losses for investors affected by alleged securities fraud between March 17, 2023 and November 4, 2024. Investors affected by the lawsuit are encouraged to request lead plaintiff status by May 20, 2025.
Sana is a biotechnology company with several drug product candidates under investigation. Amongst those drugs are SC291 in oncology; SC379 in CNS disorder treatment, and SG 299 in blood cancer treatment.
In 2023, Sana claimed it had adequate cash to fund its research through 2025. It also claimed commitment to promising product candidates SC291, SC379, and SG299, stating it was planning to submit an IND for the drugs in the coming year. Later that year, Sana began a Phase 1 trial for SC291, though it stopped its research in SG299 due to insufficient funds, so it could extend its cash runway for other programs.
In reality, Sana’s cash position was weaker than it led investors to believe. In November 2024, Sana announced it was halting its SC291 and SC379 programs to focus on type 1 diabetes drugs. It claimed doing so would extend its cash runway into 2026.
Investors reacted negatively to this news and Sana’s stock price dropped almost 10% the next day.
Sana Biotechnology, Inc. (SANA) Case Details
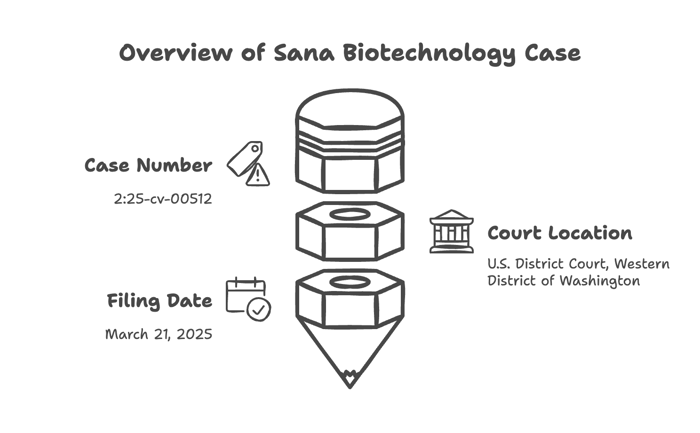
Drott. V. Sana Biotechnology, Inc., et al Case No. 2:25-cv-00512
U.S. District Court, Western District of Washington
Filed complaint on March 21, 2025
Sana Biotechnology, Inc. (SANA) Company Profile
Sana is a biotechnology company. According to its website, it “is focused on creating and delivering engineered cells as medicines for patients.” Its research focuses on treatments for B-cell mediated autoimmune diseases, treatments for CD22+ cancers, and treatments for type 1 diabetes. Sana has numerous drugs under investigation at various stages of clinical trials.
Sana’s therapies consist of in vivo and ex vivo treatments. An ex vivo treatment involves modifying cells outside the body.
Sana Biotechnology, Inc. (SANA) Securities Lawsuit Class Period
March 17, 2023 to November 4, 2024, inclusive.
Sana Biotechnology, Inc. (SANA) Class Action Lawsuit Allegations
The Complaint alleges Sana made false and misleading statements about its financial stability and its ability to advance three promising product candidates: SC291 in oncology; SC379 in CNS treatment; and SG299, an in vivo blood cancer treatment.
At the beginning of the class period (March 2023), Sana issued a press release announcing its Q4-22 and FY-22 financial results. The press release emphasized the Company’s strong cash position would enable it a runway to maintain its current operations into 2025. Sana also emphasized its commitment to and progress on its product candidates SC291, SC379, and SG299, having made progress toward an Investigatory New Drug Application (IND) on each (a major step in new drug development).
In a May 2023 press release, Sana reaffirmed its strong cash runway and announced enrollment for the Phase 1 (ARDENT) trial for SC291. The Company also iterated its commitment to file an IND for SG299 later in the year.
Sana changed its tune in October 2023, when it issued a press release announcing a strategy change. Sana revealed it would reduce funding for in vivo therapies, thus delaying the SG299 IND. It also announced it would decrease funding and cash burn and that it would keep “2024 operating cash burn . . . below $200 million.” The Company reasoned doing so would enable it to extend its current operations into 2025, at least.
Nonetheless, from November 2023 to August 2024, Sana continued to affirm its cash runway into 2025. It also continued to promote SC291, touting its progress on the ARDENT trial enrollment. In a May 2024 press release, it described the early ARDENT data as safe and showing early efficacy. The Company also published data on SC279 in a peer-reviewed journal.
Plaintiffs' filed complaint alleges these statements were deceptive, false, and misleading, and that Defendants overstated Sana's financial capacity. In reality, Sana maintained a weak cash position and lacked the resources to fund SC291 and SC279, despite its executives’ rosy statements otherwise.
The Truth Emerges
On November 4, 2024, Sana stunned investors when it issued a press release announcing it was suspending SC291 and SC379, redirecting funding into its type 1 diabetes program. The Company asserted “[w]ith these changes, Sana extends its expected cash runway into 2026.”
Market Reaction to Sana Biotechnology's Lawsuit
Investors reacted negatively to this news. Sana’s stock price fell 9.84% the next day.
Next Steps
- Submissions for lead plaintiff are due May 20, 2025.
- The Court will issue its order for lead plaintiff and counsel in the weeks after submissions are due.
- The Court will then consider motion for class certification.
- The Court will later consider a Motion to Dismiss.
To learn whether you may be eligible for a recovery under this class action, go to the case submission page found here.
![Sana Biotechnology, Inc. (SANA) Securities Class Action Lawsuit Update [April 21, 2025]](https://dropinblog.net/cdn-cgi/image/fit=scale-down,format=auto,width=700/34260815/files/featured/sana-biotechnology-lawsuit.webp)

![SoundHound AI, Inc. (SOUN) Securities Class Action Lawsuit Update [April 24, 2025]](https://dropinblog.net/cdn-cgi/image/fit=scale-down,format=auto,width=700/34260815/files/featured/soundhounc-inc-lawsuit.webp)
![Ultra Clean Holdings, Inc. (UCTT) Securities Class Action Lawsuit Update [April 30, 2025]](https://dropinblog.net/cdn-cgi/image/fit=scale-down,format=auto,width=700/34260815/files/featured/ebc77178-cda0-48a1-9240-1fd2b3d0dca6.webp)
![Zynex, Inc. (ZYXI) Securities Class Action Lawsuit Update [April 21, 2025]](https://dropinblog.net/cdn-cgi/image/fit=scale-down,format=auto,width=700/34260815/files/featured/zynex-lawsuit-1.webp)