Introduction to Unicycive Therapeutics, Inc. (UNCY) Securities Class Action Lawsuit
A federal securities fraud class action has been filed against Unicycive Therapeutics, Inc. (NASDAQ: UNCY) covering March 29, 2024 through June 27, 2025. Investors allege the company overstated its readiness to meet FDA manufacturing compliance requirements and exaggerated the regulatory prospects of its NDA for oxylanthanum carbonate (OLC). During June 2025, the FDA identified cGMP deficiencies at a third-party manufacturing vendor and later issued a Complete Response Letter (CRL) citing those deficiencies. On these revelations, Unicycive's stock fell sharply on two separate trading days. The complaint was filed on August 15, 2025, in the U.S. District Court, Northern District of California.
Unicycive Therapeutics, Inc. (UNCY) Securities Lawsuit Case Details
Case Name: Elkhodari v. Unicycive Therapeutics, Inc., et al.
Case No.: 3:25-cv-06923
Jurisdiction: U.S. District Court, Northern District of California
Filed on: August 15, 2025
Unicycive Therapeutics, Inc. (UNCY) Company Profile
Unicycive is a clinical-stage biotechnology company developing therapies to address unmet medical needs in the U.S. The Company is developing, among other therapies, oxylanthanum carbonate (OLC), a purported next‑generation phosphate binder candidate for treating hyperphosphatemia in chronic kidney disease patients on dialysis.
Unicycive Therapeutics, Inc. (UNCY) Securities Lawsuit Class Period
March 29, 2024–June 27, 2025, inclusive.
All those who purchased or otherwise acquired Unicycive securities during the Class Period and were damaged upon the revelation of the alleged corrective disclosures may be eligible to join the Unicycive Therapeutics, Inc. (UNCY) class action lawsuit. Defendants, the officers and directors of the Company at all relevant times, members of their immediate families and their legal representatives, heirs, successors or assigns, and any entity in which Defendants have or had a controlling interest are excluded.
Allegations in the Unicycive Therapeutics, Inc. (UNCY) Securities Class Action Lawsuit
The lawsuit names Unicycive, CEO Shalabh Gupta, and CFO John Townsend. According to the complaint, they presented OLC as on track for approval and launch while assuring investors about manufacturing readiness and regulatory momentum.
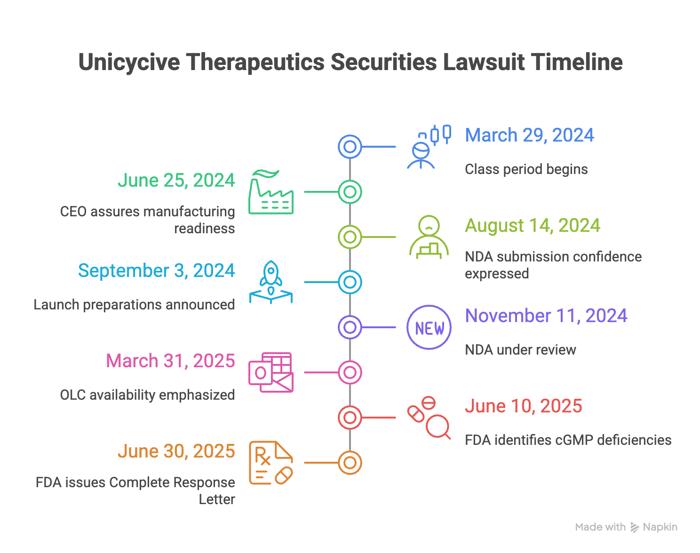
Just before the class period began, on March 28, 2024, Gupta said positive trial results would support an NDA filing with the FDA, with topline data expected late in the second quarter and an NDA to be filed shortly thereafter. As the period progressed, on June 25, 2024, he told investors on a conference call that manufacturing was "very, very much ready," with validated batches at "multi hundreds of kilograms," that there was "no problem," and that the company was "ready to launch mid next year."
Through the back half of 2024, Unicycive reinforced this message, as alleged in the complaint. On August 14, 2024, Gupta stated the company "completed all the necessary requirements" from the pivotal trial to fulfill the FDA's requests and remained on track to submit the NDA by month's end, expressing "a high degree of confidence" in OLC's potential if approved. On September 3, 2024, he said the team was "preparing to launch OLC, if approved," and on November 11, 2024, he added that with the "NDA now under review," Unicycive was preparing to commercialize and launch OLC in the second half of 2025. On March 31, 2025, he again emphasized preparations to "rapidly make OLC available to patients upon approval."
The complaint alleges that, despite these assurances, Unicycive's readiness and ability to satisfy the FDA's manufacturing compliance requirements was overstated and that the OLC NDA's regulatory prospects were likewise overstated, rendering the public statements false and misleading at all relevant times.
The Truth Emerges
On June 10, 2025, Unicycive disclosed that the FDA had identified deficiencies in cGMP compliance at a third-party manufacturing vendor—specifically, a subcontractor of its CDMO and not its drug substance vendor—following an FDA inspection. The company added that, given these deficiencies, any label discussions with the FDA were precluded.
Days after the class period ended on June 27, 2025, additional FDA-related developments were disclosed. On June 30, 2025, Unicycive announced the FDA had issued a CRL for the OLC NDA to treat hyperphosphatemia in CKD patients on dialysis, citing the same cGMP deficiencies at the third‑party subcontractor. According to the complaint, these developments allegedly showed that prior assurances about manufacturing readiness and the NDA's regulatory prospects were overstated.
Market Reaction
Following each pre-market disclosure, Unicycive's stock declined that same trading day. On June 10, 2025, following the disclosure of FDA‑identified cGMP deficiencies and the halt to label discussions, Unicycive's stock fell $3.68 per share, or 40.89%, to close at $5.32, with the news disseminated in pre‑market hours.
Momentum turned further when, on June 30, 2025, after Unicycive announced the FDA's CRL citing the same deficiencies, the stock declined another $2.03 per share, or 29.85%, to close at $4.77, again following a pre‑market announcement.
Next Steps
- Submissions for lead plaintiff are due October 14, 2025.
- The Court will issue its order for lead plaintiff and counsel in the weeks after submissions are due.
- The Court will then consider motion for class certification.
- The Court will later consider a Motion to Dismiss.
To learn if you are eligible for recovery under the UNCY securities class action lawsuit, visit the case submission page here.
Disclaimer: This shareholder alert is for informational purposes only and does not constitute legal advice. Consult a qualified attorney for personalized guidance. No specific outcomes are guaranteed.
![Unicycive Therapeutics, Inc. (UNCY) Securities Class Action Lawsuit Update [September 30, 2025]](https://dropinblog.net/cdn-cgi/image/fit=scale-down,format=auto,width=700/34260815/files/featured/uncy-therapeutics-class-action-blog-banner.webp)


![Charter Communications, Inc. (CHTR) Securities Class Action Lawsuit Update [September 30, 2025]](https://dropinblog.net/cdn-cgi/image/fit=scale-down,format=auto,width=700/34260815/files/featured/charter-communications-chtr-securities-lawsuit-blog-banner.webp)
![Snap Inc. (SNAP) Securities Class Action Lawsuit Filed [September 30, 2025]](https://dropinblog.net/cdn-cgi/image/fit=scale-down,format=auto,width=700/34260815/files/featured/20251003-1114-snap-securities-alert-simple-compose-01k6na4208evatnxkgx0wd3rg2.png)
