AMPE Lawsuit Filed; Levi & Korsinsky Announces AMPE Class Action
Levi & Korsinsky, LLP
September 12, 2018
Shi v. Ampio Pharmaceuticals, Inc. et al 2:18-cv-07476 — On August 25, 2018, investors sued Ampio Pharmaceuticals, Inc. (Ampio, AMPE or the Company) in United States District Court, Central District of California. Plaintiffs in the AMPE class action allege that they acquired Ampio stock at artificially inflated prices between December 14, 2017 and August 7, 2018 (the “Class Period”). They are now seeking compensation for financial losses incurred upon public revelation of the Company’s alleged misconduct during that time. For more information about the AMPE Lawsuit, please contact us today!
Summary of the Allegations
Company Background
Ampio (NYSE: AMPE) bills itself as an “innovative drug discovery and development company” that engages in the development of medicinal treatments for inflammatory diseases, including but not limited to osteoarthritis.
The Company claims that its, “therapeutic product pipeline has been developed through more than two decades of study at leading hospital-based research centers.” It also claims that it has received more than 100 patents globally and that it has “hundreds more” pending.
Ampio’s contentions about one of its lead product candidates, Ampion, are at the crux of the August 25 lawsuit.
Summary of Facts
Ampio and two of its senior officers and directors (the “Individual Defendants”) are now accused of deceiving investors by lying and withholding critical information about the Company’s business practices and prospects during the Class Period.
Specifically, they are accused of omitting truthful information about the status of a Phase 3 clinical trial for Ampion from SEC filings and related material. By knowingly or recklessly doing so, they allegedly caused Ampio stock to trade at artificially inflated prices during the time in question.
The truth emerged after the market closed on August 7, 2018. That’s when the Company announced “updated business disclosures” in which it disclosed that the FDA had not approved one of its clinical trials for Ampion. It said in pertinent part, “Despite our belief that the APC-003-C trial design was based on FDA guidance and feedback consistent with FDA precedent for similar products (in intended use, in origin, and in regulatory pathway), which we reiterated with the FDA multiple times, the FDA does not consider the AP-003-C [sic] trial to be an adequate and well-controlled clinical trial.”
A closer look…
As alleged in the August 25 complaint, the Company and/or Individual Defendants repeatedly made false and misleading public statements during the Class Period.
For example, in a press release issued at the beginning of the Class Period, the Company said in pertinent part: “We look forward to working closely with the U.S. Food and Drug Administration (FDA) as we prepare to submit our Biologics License Application (BLA) for Ampion.”
Then, on a “slide deck” attached to a form filed with the SEC on January 8, 2018, the Company said in pertinent part: “Ampion has successfully completed two pivotal Phase 3 trials for the signs and symptoms [sic] severe OAK.” (Osteoarthritis of the knee.)
Finally, on a form filed with the SEC on March 6, 2018, the Company said in pertinent part: “When treated with Ampion, patients experienced significant improvement in a composite endpoint of pain and function compared to all severely diseased saline-treated patients in historical Ampion phase II clinical trials.”
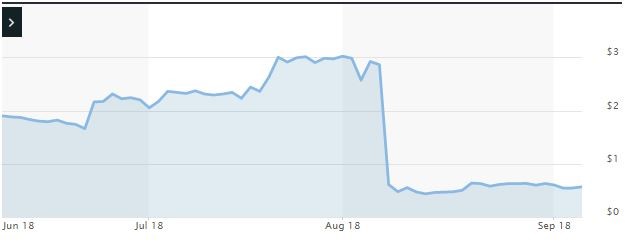
Impact of the Alleged Fraud on Ampio’s Stock Price and Market Capitalization
| Closing stock price prior to disclosures:
|
$2.86 |
| Closing stock price the trading day after disclosures:
|
$0.61 |
| One day stock price decrease (percentage) as a result of disclosures:
|
78.67% |
The following chart illustrates the stock price during the class period:
Actions You May Take
If you have purchased shares during the Class Period, you may join the class action as a lead plaintiff, remain a passive class member, or opt out of this litigation and pursue individual claims that may not be available to the class as a whole.
NOTE: The deadline to file for lead plaintiff in this class action is October 24, 2018. You must file an application to be appointed lead plaintiff prior to this deadline in order to be considered by the Court. Typically, the plaintiff or plaintiffs with the largest losses are appointed lead plaintiff.
In order to identify your potential exposure to the alleged fraud during the time in question, you may wish to perform an analysis of your transactions in Ampion common stock using court approved loss calculation methods.
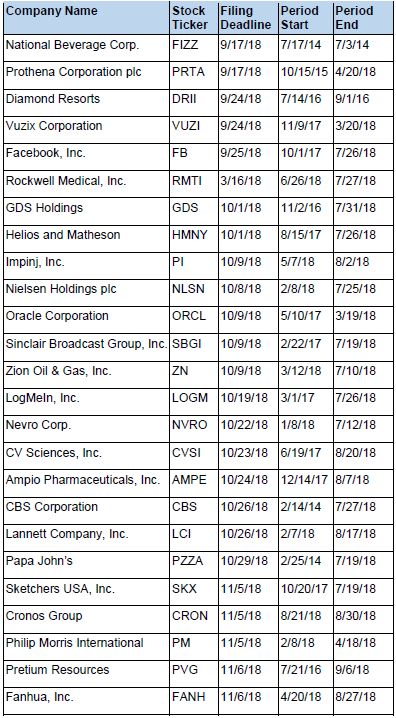
Recently Filed Cases
Listed below are recently filed securities class action cases being monitored by us, along with the class period and the deadline to file a motion to be appointed as the Lead Plaintiff in the action. Please contact us if you would like an LK report for any of these cases:
About Us
Levi & Korsinsky is a leading securities litigation firm with a hard-earned reputation for protecting investors’ rights and recovering losses arising from fraud, mismanagement and corporate abuse. With thirty attorneys and offices in New York, Connecticut, California and Washington D.C., the firm is able to litigate cases in various jurisdictions in the U.S., England, and in other international jurisdictions.
Levi & Korsinsky provides portfolio monitoring services for high-net worth investors and institutional clients. Our firm also assists investors in evaluating whether to opt-out of large securities class actions to pursue individual claims.
For additional information about this case or our institutional services, please contact us.