FLKS Class Action Report
Levi & Korsinsky, LLP
July 2, 2018
On June 19, investors sued Flex Pharma, Inc. (“Flex Pharma” or the “Company”) in United States District Court, Southern District of New York. Plaintiffs in the federal securities class action allege that they acquired Flex Pharma stock at artificially inflated prices between November 6, 2017, and June 12, 2018. They are now seeking compensation for financial losses incurred upon public revelation of the Company’s alleged misconduct during that time. Here’s what you need to know about the Flex class action (FLKS class action):
Summary of the Allegations
Company Background
Flex Pharma (NASDAQ; FLKS) is clinical-stage biotechnology company. As such, it develops “innovative and proprietary treatments for cramps and spasticity.”
Specifically, the Company creates medicines that can be used to treat ailments ranging from nighttime leg cramps to “cervical dystonia, spinal cord spasticity and multiple sclerosis.”
The Company’s claims about its lead product candidate, FLX-787, which was evaluated as a potential treatment for amyotrophic lateral sclerosis (“ALS”) and Charcot-Marie-Tooth disease (“CMT”), are at the crux of the current lawsuit.
Summary of Facts
Flex Pharma and two of its senior officers and/or directors (the “Individual Defendants”) now stand accused of deceiving investors by lying and withholding critical information about the Company’s business, operational and compliance policies during the Class Period.
Specifically, they are accused of omitting truthful information about its lead product candidate from SEC filings and related material. By knowingly or recklessly doing so, they allegedly caused Flex Pharma stock to trade at artificially inflated prices during the time in question.
The truth came out when Flex Pharma issued a press release on June 13, 2018. In it, the Company announced that it would end “its ongoing Phase 2 clinical trial investigations of FLX-787 in amyotrophic lateral sclerosis (ALS) and Charcot-Marie-Tooth disease (CMT) due to oral tolerability concerns in both studies.”
A closer look…
As alleged in the June 19 complaint, Flex Pharma repeatedly made misleading public statements during the Class Period.
For example, on a form filed with the SEC at the beginning of the Class Period, the Company said in pertinent part: “Our development programs are steadily advancing. We have initiated our Phase 2b ALS trial under Fast Track designation, and more recently, our Phase 2b CMT trial. These two studies, as well as the ongoing exploratory spasticity study in MS in Australia are expected to yield several important data readouts in 2018.”
On another form filed with the SEC on March 7, 2018, Flex Pharma stated in pertinent part: “The next 12 months will be transformational for Flex as we expect to report results from a number of larger clinical trials – first an exploratory spasticity study in MS, followed by two Phase 2 studies in ALS and a Charcot-Marie-Tooth patients, with our current cash position taking us to mid 2019.”
Finally, on another form filed with the SEC on May 2, 2018, the Company said on pertinent part: “The past few months have been particularly rewarding on the clinical front, as we achieved significant milestones with positive data in two serious and distinctly different neurological diseases: MS and ALS. We believe these data demonstrate the clear potential of FLX-787 to reduce painful crams and spasms in these patient populations.”
Impact of the Alleged Fraud on Flex Pharma’s Stock Price and Market Capitalization
| Closing stock price prior to disclosures:
|
$4.18 |
| Closing stock price the trading day after disclosures:
|
$1.04 |
| One day stock price decrease (percentage) as a result of disclosures:
|
75.12% |
The following chart illustrates the stock price during the class period:
Actions You May Take
If you have purchased shares during the Class Period, you may join the class action as a lead plaintiff, remain a passive class member, or opt out of this litigation and pursue individual claims that may not be available to the class as a whole.
NOTE: The deadline to file for lead plaintiff in this class action is August 20, 2018. You must file an application to be appointed lead plaintiff prior to this deadline in order to be considered by the Court. Typically, the plaintiff or plaintiffs with the largest losses are appointed lead plaintiff.
In order to identify your potential exposure to the alleged fraud during the time in question, you may wish to perform an analysis of your transactions in Flex Pharma common stock using court approved loss calculation methods.
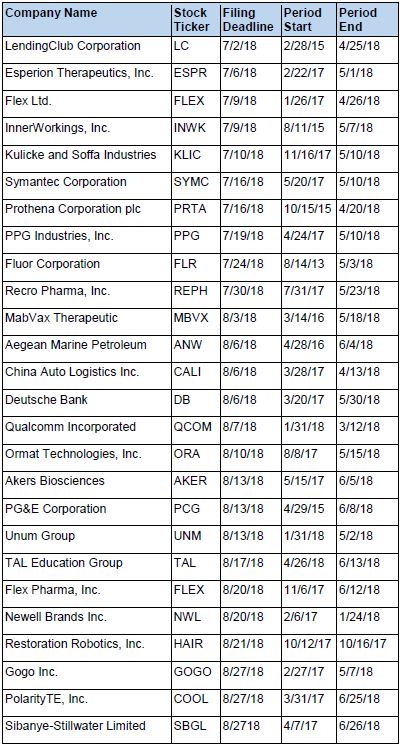
Recently Filed Cases
Listed below are recently filed securities class action cases being monitored by us, along with the class period and the deadline to file a motion to be appointed as the Lead Plaintiff in the action. Please contact us if you would like an LK report for any of these cases:
About Us
| This information is provided for general information purposes only, and should not be construed as legal advice, nor does it establish an attorney-client relationship with Levi & Korsinsky LLP. Any and all information herein is simply an opinion based on publicly available information and should not necessarily be construed as fact. For more information, please visit our website at www.zlk.com.
Attorney Advertising
|
Levi & Korsinsky is a leading securities litigation firm with a hard-earned reputation for protecting investors’ rights and recovering losses arising from fraud, mismanagement and corporate abuse. With thirty attorneys and offices in New York, Connecticut, California and Washington D.C., the firm is able to litigate cases in various jurisdictions in the U.S., England, and in other international jurisdictions.
Levi & Korsinsky provides portfolio monitoring services for high-net worth investors and institutional clients. Our firm also assists investors in evaluating whether to opt-out of large securities class actions to pursue individual claims.
For additional information about this case or our institutional services, please contact us.