Introduction to Actinium Pharmaceuticals, Inc. (ATNM) Lawsuit
This is a shareholder alert about the Actinium Pharmaceuticals, Inc. class action lawsuit filed on or about March 27, 2025. The class action seeks to recover losses of shareholders who were adversely affected by alleged securities fraud between October 31, 2022 and August 2, 2024. The lawsuit alleges that the defendants made false and misleading statements about Actinium's Iomab-B drug and its FDA approval likelihood. Investors who purchased Actinium securities during the class period may be entitled to compensation without out-of-pocket fees through a contingency fee arrangement. Investors have until May 27, 2025 to file submissions as lead plaintiff.
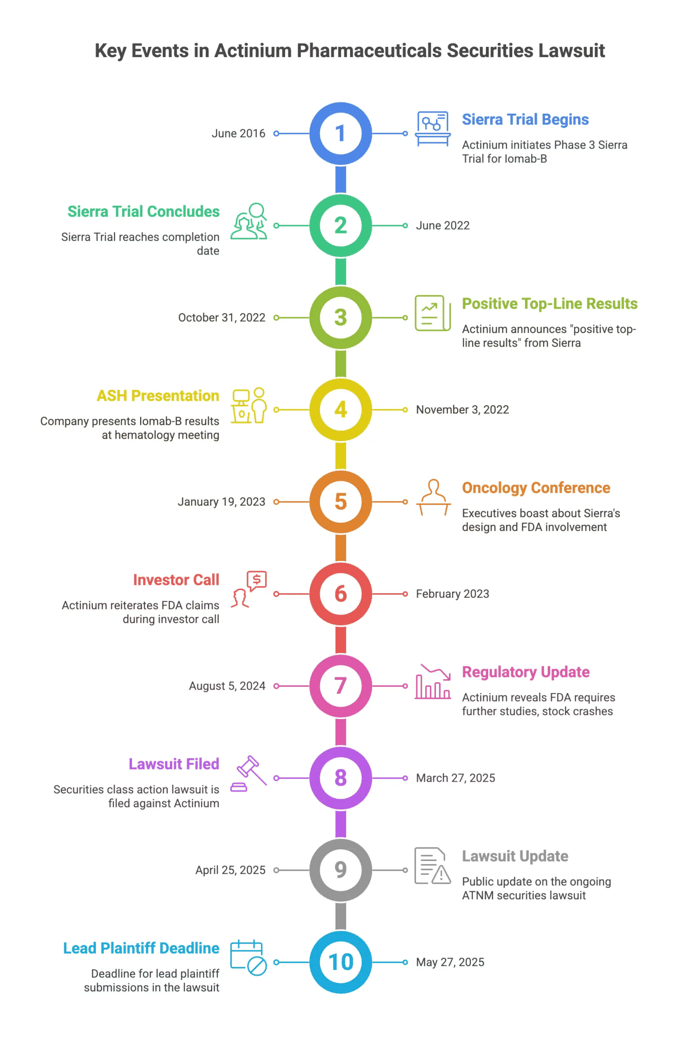
Actinium is a biopharmaceutical company. One of its primary products is Iomab-B, a cancer-fighting agent. Actinium launched the Sierra Trial to study Iomab-B’s effectiveness. Sierra used a non-conventional study design; however, Actinium told investors the FDA both approved and assisted in creating this design.
In 2022, Actinium completed the Sierra study and boasted about Iomab-B’s success, claiming the drug was set for FDA approval. In August 2024, the Company provided a regulatory update regarding Iomab-B. Actinium revealed the FDA's guidelines now required the Company complete extensive new drug testing, using conventional study methods. These revelations undercut Actinium’s previous claims that the FDA blessed its unusual study plan.
Investors reacted negatively: Actinium’s stock price crashed 60% in one day.
Actinium Pharmaceuticals, Inc. (ATNM) Case Details
Kohil v. Actinium Pharmaceuticals, Inc., et al Case No. 1:25-cv-02553
U.S. District Court, Southern District of New York
Filed on March 27, 2025
Actinium Pharmaceuticals, Inc. (ATNM) Company Profile
Actinium is a biopharmaceutical company that develops radiotherapies. One of its products, Iomab-B, is used before bone marrow transplants and might be used certain forms of myeloid leukemia for people who failed traditional treatments.
Actinium Pharmaceuticals, Inc. (ATNM) Securities Lawsuit Class Period
October 31, 2022 to August 2, 2024, inclusive.
Investors who acquired Actinium securities during the relevant time frame might be eligible to join the Actinium pharmaceuticals lawsuit.
Actinium Pharmaceuticals, Inc. (ATNM) Lawsuit Allegations
In June 2016, Actinium initiated the Phase 3 Sierra Trial evaluating Iomab-B. Unlike many clinical trials, Sierra was not double-blinded. Actinium claimed the FDA assisted in planning and designing Sierra. The study completed in June 2022.
On October 31, 2022, Actinium issued a press release announcing Sierra purportedly demonstrated “positive top-line results," including improved overall survival. The Company said these results moved Iomab-B toward FDA approval. On November 3, 2022, the Company announced a presentation at the Annual American Society of Hematology Meeting, touting Iomab-B’s success.
On January 19, 2023, Actinium executives boasted about the Company's data at an oncology conference. Executives bragged about Sierra’s “pristine” design and even boasted about the FDA’s role in the design. They said “it’s an experiment that really the FDA set up in conjunction with us[.]” The Company reiterated these claims about the FDA during a February 2023 investor call.
The filed complaint alleges Actinium made false statements about the Sierra Trial design and its claims about the Iomab-B results were deceptive, false, and misleading. Plaintiffs claim the Sierra data was unlikely to meet FDA guidelines; the Iomab-B data was not as glowing as Actinium originally claimed; and, as a result, the FDA was unlikely to approve Iomab-B. Plaintiffs assert Defendants' positive statements during the class period about Sierra and Iomab-B artificially inflated Actinium's stock price; once the truth emerged, Actinium's stock price plunged, costing investors millions.
The Truth Emerges
The truth emerged on August 5, 2024 when Actinium published a regulatory update. In it, Actinium revealed that despite Sierra meeting its primary endpoint, “the FDA has now determined that demonstrating an overall survival benefit in a randomized head-to-head trial is required[.]” The FDA advised Actinium of extensive further studies required prior to FDA approval of Iomab-B.
Market Reaction
Investors reacted negatively: Actinium’s stock price plunged 60% in one day.
Next Steps
Submissions for lead plaintiff are due May 27, 2025.
The Court will issue its order for lead plaintiff and counsel in the weeks after submissions are due.
The Court will then consider motion for class certification.
The Court will later consider a Motion to Dismiss.
To learn whether you may be eligible for a recovery under this class action, go to the case submission page found here.
![Actinium Pharmaceuticals, Inc. (ATNM) Securities Class Action Lawsuit Update [April 25, 2025]](https://dropinblog.net/cdn-cgi/image/fit=scale-down,format=auto,width=700/34260815/files/featured/atnm.jpeg)

![Canopy Growth Corp. (CGC) Securities Class Action Lawsuit Update [May 5, 2025]](https://dropinblog.net/cdn-cgi/image/fit=scale-down,format=auto,width=700/34260815/files/featured/cgc-sh-alert.jpeg)
![Solaris Energy Infrastructure, Inc. (SEI) Securities Class Action Lawsuit Update [April 30, 2025]](https://dropinblog.net/cdn-cgi/image/fit=scale-down,format=auto,width=700/34260815/files/featured/sei.jpeg)
