Introduction to Biohaven Ltd. (BHVN) Securities Class Action Lawsuit
Biohaven Ltd. (NYSE: BHVN) has been hit with a securities class action lawsuit. The case represents investors who purchased or otherwise acquired Biohaven securities between March 24, 2023 and May 14, 2025 (the “Class Period”). The complaint alleges Biohaven and some of its executives ran afoul of federal securities laws based on materially false and misleading statements made about the company's business prospects.
Plaintiffs say Biohaven and some of its executives misled investors about the efficacy and clinical prospects of two of its drugs’ regulatory prospects. Those drugs are troriluzole (for spinocerebellar ataxia, "SCA") and BHV-700 (for bipolar).
From July 2023 to May 2025, the Company made a series of statements touting both drugs’ efficacy and chances for FDA approval. But after lackluster trial results, the FDA balked. Biohaven’s stock price plunged. 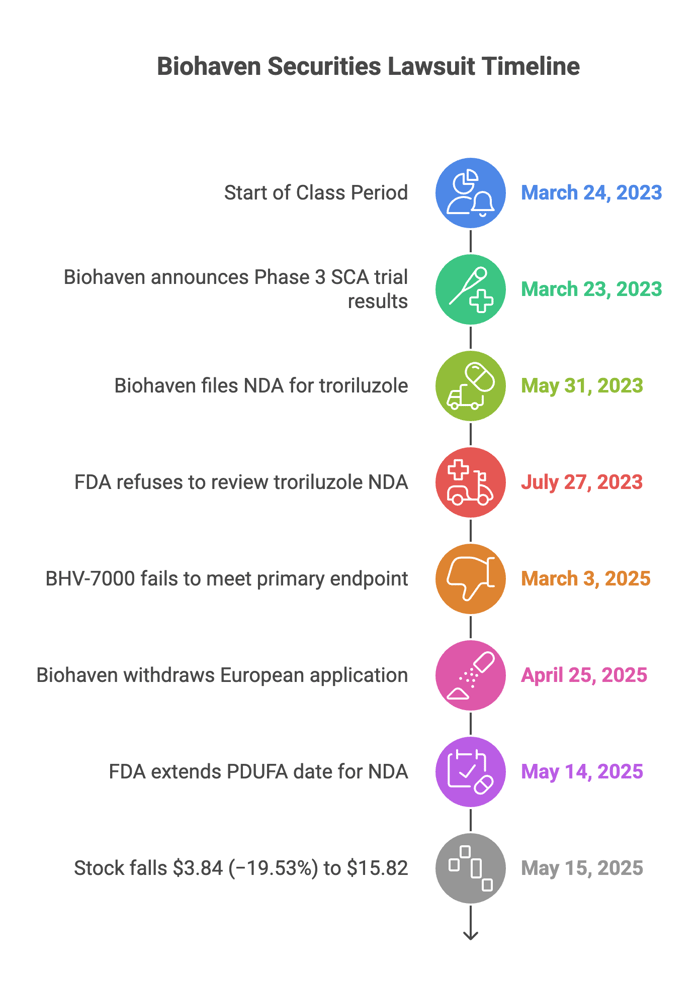
Biohaven Ltd. (BHVN) Securities Lawsuit Case Details
- Caption: Taylor v. Biohaven Ltd., et al.
- Docket Number: 3:25-cv-01120
- Court: U.S. District Court, District of Connecticut
- Filing Date: July 14, 2025
Biohaven Ltd. (BHVN) Company Profile
Biohaven (NYSE: BHVN) is a Connecticut-based biopharmaceutical company. It focuses on therapies in immunology, neuroscience, and onology. During the Class Period, Biohaven tested troriluzole for SCA and BHV-7000 for bipolar disorder.
Biohaven Ltd. (BHVN) Lawsuit Class Period
March 24, 2023 – May 14, 2025
People who bought Biohaven securities during the class period may might be eligible to join the Biohaven securities class action lawsuit. 
Allegations in the Biohaven Ltd. (BHVN) Securities Class Action Lawsuit
From March 2023 to early 2025, Biohaven issued a variety of public statements – such as press releases, SEC filings, and investor calls. Those statements boasted troriluzole showed “clinically meaningful improvement” in SCA patients. Executives also said BHV-7000 had a “favorable clinical profile.”
On March 23, 2023, Biohaven said its Phase 3 SCA trial on troriluzole showed great results and execs hyped troriluzole's regulatory prospects. On May 31, 2023, a press release announced Biohaven submitted its troriluzole New Drug Application ("NDA," filed with the FDA).
But the FDA submission didn’t go as planned. On July 27, 2023, Biohaven admitted the FDA flat-out refused to review the NDA. The government said troriluzole didn’t meet the primary endpoint. Despite the FDA’s pushback, the company continued to promote troriluzole’s regulatory approval prospects and BHV-7000’s efficacy.
Throughout 2024, Biohaven repeatedly claimed it was having “constructive dialogue” with the Agency. It also claimed it was trying again and announced a December 2024 resubmission of the troriluzole NDA. The company also reported topline results from a real-world evidence study and stated that the FDA's office had accepted the resubmitted NDA for Priority Review in February 2025.
Plaintiffs say these statements overstated the data’s strength. They also claim defendants' public statements misstated the changes for regulatory approval and misrepresented the clinical prospects of both drug candidates. These statements, in turn, caused Biohaven’s stock price to artificially climb – before ultimately crashing when the truth came out.
The Truth Emerges
Plaintiffs claim that the truth about troriluzole and BHV-7000 came out as a slow trickle from 2023 to 2025. They point to several key events:
- July 27, 2023: FDA refusal to review the initial troriluzole NDA.
- March 3, 2025: Admission that BHV-7000 didn’t meet its primary endpoint in a major study.
- April 25, 2025: News reports confirmed that Biohaven had withdrawn its European application for troriluzole.
- May 14, 2025: Biohaven announced that the FDA extended the PDUFA date for the resubmitted NDA by three months.
Market Reaction
After the first bad news about the FDA refusal, through to May 2025, Biohaven’s stock dropped again and again:
- July 27, 2023: Stock fell $5.38 (−22.61%) to $18.42
- March 3, 2025: Stock fell $5.12 (−13.77%) to $32.06
- April 25, 2025: Stock fell $3.56 (−15.21%) to $19.84
- May 15, 2025: Stock fell $3.84 (−19.53%) to $15.82
Next Steps
- Lead Plaintiff Deadline: Submissions to serve as lead plaintiff are due September 12, 2025
- The Court will determine lead plaintiff and counsel.
- Procedural steps may include motions to dismiss and class certification.
To learn if you are eligible for recovery under the Biohaven class action lawsuit, visit the case submission page here.
Disclaimer: This alert is based solely on allegations contained in the filed complaint and publicly available information. It is provided for informational purposes only and does not constitute legal advice. No outcome is guaranteed. Investors should consult their own counsel regarding rights or potential claims.


![Petco Health and Wellness Company, Inc. (WOOF) Securities Class Action Lawsuit Update [August 12, 2025]](https://dropinblog.net/cdn-cgi/image/fit=scale-down,format=auto,width=700/34260815/files/featured/petco-securities-class-action-lawsuit-blog-banner.webp)
![Centene Corporation (CNC) Securities Class Action Lawsuit Update [August 18, 2025]](https://dropinblog.net/cdn-cgi/image/fit=scale-down,format=auto,width=700/34260815/files/featured/cnc-banner-imagewebp.png)
