Introduction to Iovance Biotherapeutics lawsuit
A securities fraud class action lawsuit has been filed against Iovance Biotherapeutics, Inc. (NASDAQ: IOVA) on behalf of investors who purchased the Company’s common stock between August 8, 2024 and May 8, 2025 (the “Class Period”). The Iovance securities fraud lawsuit alleges that Iovance and certain executives violated federal securities laws, including the Securities Exchange Act, by making false and misleading statements regarding the company’s revenue guidance, treatment center readiness, and commercial launch capabilities for its flagship product, Amtagvi.

Iovance Biotherapeutics (IOVA) Lawsuit Case Details
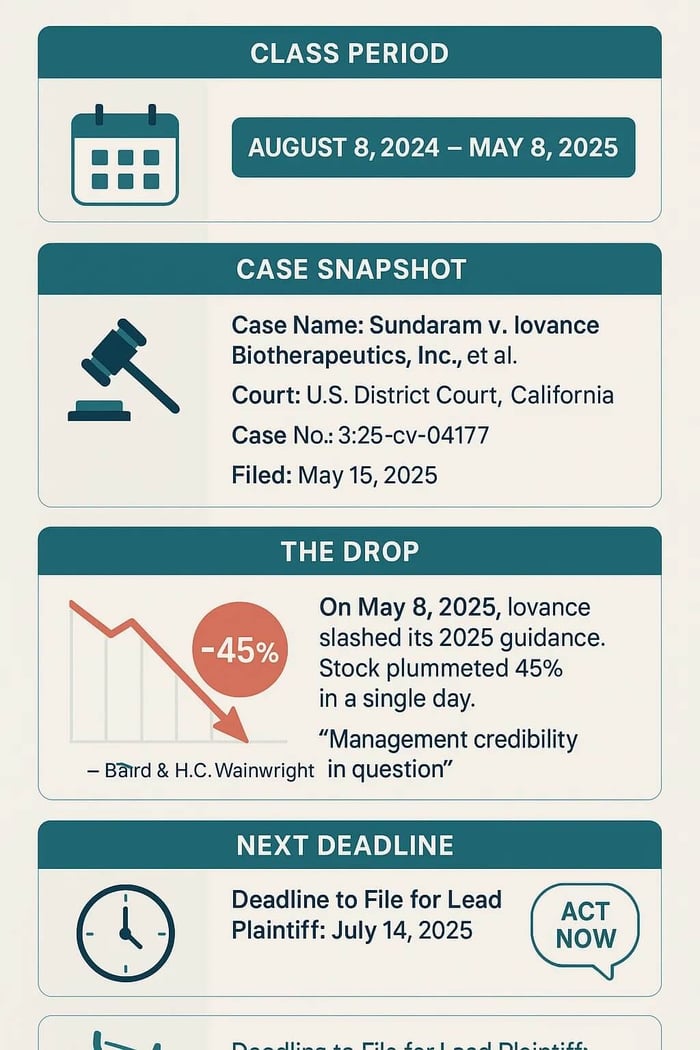
Sundaram v. Iovance Biotherapeutics, Inc., et al
Case No.: 3-25-cv-04177
United States District Court, Northern District of California
Filed on May 15, 2025
Iovance Biotherapeutics (IOVA) Company Profile
Iovance Biotherapeutics is a commercial-stage biopharmaceutical company. Its business is focused on developing and commercializing autologous cell therapies for solid tumor cancers. Its lead product its Amtagvi. Amtagvi is an autologous T-cell immunotherapy treatment for metastatic melanoma. The company also distributes Proleukin, an interleukin-2 product used in conjunction with Amtagvi. Treatments are administered through the Company's Authorized Treatment Centers (ATCs) across the U.S.
Class Period:
August 8, 2024 to May 8, 2025, inclusive.
Investors who purchased Iovance securities during the class period might be eligible to join the Iovance Biotherapeutics class action lawsuit.
Allegations in the Iovance Biotherapeutics (IOVA)
Plaintiffs allege Iovance repeatedly issued overly optimistic financial guidance and other public statements that materially overstated the Company’s commercial progress on Amtagvi. Specifically, the Complaint alleges Iovance misrepresented its ability to scale up its operations through its ATC network and manufacture enough product to meet projected demand.
On August 8, 2024, Iovance announced its Q2-2024 earnings report and projected Fiscal Year 2024 revenue projections. The Company expected $450-$475 million in product revenue for fiscal year 2025. Iovance reiterated that figure in its Q3-2024 earnings press release, issued November 7, 2024. In that same press release, Iovance assured investors of strong Amtagvi adoption and that its ATC network was positioned to support great growth. During the associated earnings call, the Company’s CEO and COO both touted the rollout of new treatment centers, growing demand for Amtagvi, and reiterated the Iovance’s glowing guidance for 2025.
The 2024 Form 10-K, filed with the SEC on February 27, 2025, doubled down on this narrative. Iovance reported $161.4 million in full-year product revenue, with Amtagvi accounting for the vast bulk of that revenue. Importantly, the Company maintained its $450-$475 million revenue projection for 2025 in this filing. The Company also claimed more than 200 patients had been treated and more than 70 ATCs were online – both of which pointed toward accelerating growth.
But, the Complaint says these statements deceived investors and were materially false and misleading. Plaintiffs allege Iovance’s public statements omitted critical facts: the Company was facing major difficulties scaling up its ATC infrastructure, many treatment center had not treated any patients, and others were only treating very small numbers of patients. The Complaint alleges that these misstatements and omissions propped up Iovance’s stock and caused investors to purchase Iovance shares at artificially high prices. Once the truth was revealed, and Iovance’s stock price plummeted, those investors suffered heavy losses.
The Truth Emerges
The truth came out on May 8, 2025 when Iovance published its Q1 2025 financial results and hosted its associated earnings call. The Company reported $49.3 million in total revenue – a sharp decline from the prior quarter. It also shocked investors when it slashed its fiscal 2025 guidance to a new range of $250-$300 million. Executives offered several excuses for the guidance reduction. The Company blamed a weaker-than-expected ATC ramp-up, lower-than-projected manufacturing success rates, and delays in scaling community referral programs. He also said the forecast was based on flawed modeling.
Market Reaction
Investors reacted negatively to this news and Iovance’s stock price fell 45% in a single day. Analysts also expressed surprise at Iovance’s stunning lack of credibility. Coverage from Baird and H.C. Wainwright discussed management’s credibility gap and the underperformance of the ATC network as key concerns.
Next Steps
Submissions for lead plaintiff are due July 14, 2025.
The Court will issue its order for lead plaintiff and counsel in the weeks after submissions are due.
The Court will then consider motion for class certification.
The Court will later consider a Motion to Dismiss.
To learn if you are eligible for recovery under the Iovance Biotherapeutics securities class action lawsuit, visit the case submission page here.
Disclaimer: This shareholder alert is for informational purposes only and does not constitute legal advice. Consult a qualified attorney for personalized guidance. No specific outcomes are guaranteed.
![Iovance Biotherapeutics (IOVA) Securities Class Action Lawsuit Update [June 13, 2025]](https://dropinblog.net/cdn-cgi/image/fit=scale-down,format=auto,width=700/34260815/files/featured/iovance-lawsuit-blog-banner.webp)

![MicroStrategy Inc. (MSTR) Securities Class Action Lawsuit Update [June 14, 2025]](https://dropinblog.net/cdn-cgi/image/fit=scale-down,format=auto,width=700/34260815/files/featured/microstrategy-securities-lawsuit-blog-banner.webp)

![Krispy Kreme, Inc. (DNUT) Securities Class Action Lawsuit Update [June 11, 2025]](https://dropinblog.net/cdn-cgi/image/fit=scale-down,format=auto,width=700/34260815/files/featured/krispy-kreme-class-action-lawsuit.webp)