Introduction to Savara Inc. (SVRA) Securities Class Action Lawsuit
A federal securities class action has been filed against Savara Inc. (NASDAQ: SVRA) in the U.S. District Court for the Eastern District of Pennsylvania. The lawsuit covers investors who acquired Savara securities between March 7, 2024 and May 23, 2025, both dates inclusive. Investors allege the Company misrepresented the readiness and timeline of its Biologics License Application (BLA) for MOLBREEVI, including whether it contained sufficient Chemistry, Manufacturing, and Controls (CMC) information and the likelihood of timely FDA approval. The story turned on May 27, 2025, when Savara disclosed an FDA Refusal to File letter stating the March 2025 BLA was not sufficiently complete and needed more CMC data. On this news, the stock fell sharply, and plaintiffs claim significant losses followed.
Savara Inc. (SVRA) Securities Lawsuit Case Details
Case Name: Ho v. Savara Inc., et al.
Case No.: 2:25-cv-05147
Jurisdiction: U.S. District Court, Eastern District of Pennsylvania
Filed on: September 8, 2025
Savara Inc. (SVRA) Company Profile
Savara is a clinical-stage biopharmaceutical company focused on rare respiratory diseases. Its lead product candidate is MOLBREEVI (molgramostim), an inhaled GM-CSF being developed for autoimmune pulmonary alveolar proteinosis (aPAP) and evaluated in the Phase 3 IMPALA-2 pivotal trial.
Savara Inc. (SVRA) Securities Lawsuit Class Period
March 7, 2024–May 23, 2025, inclusive
Eligible investors include all persons and entities other than Defendants that purchased or otherwise acquired Savara securities during this period and may be eligible to join the Savara Inc. (SVRA) class action lawsuit.
Allegations in the Savara Inc. (SVRA) Securities Class Action Lawsuit
According to the complaint, Savara Inc., Chief Executive Officer Matthew Pauls, and Chief Financial Officer David Lowrance told investors throughout 2024 and early 2025 that the Company was on track to file and complete a rolling BLA for MOLBREEVI within a tight timetable. They allegedly framed the regulatory path as clear and imminent, tying the filing to positive IMPALA-2 results and signaling a near-term U.S. approval if priority review were granted.
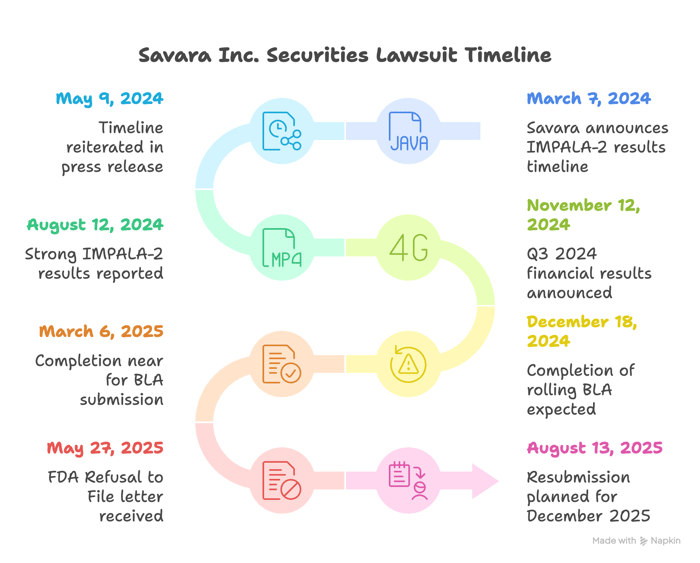
The narrative began on March 7, 2024, when Pauls said in a press release that the Company looked forward to reporting IMPALA-2 top-line results by the end of the second quarter and, assuming positive data, anticipated filing the BLA in the first half of 2025. This timeline was repeated in another press release on May 9, 2024. On August 12, 2024, after reporting strong IMPALA-2 results, Pauls stated that Savara planned to complete the BLA submission in the first half of 2025.
On November 12, 2024, the Company issued a press release announcing Q3 2024 financial results. In that release it was announced that following a pre-BLA meeting with the FDA, Savara would initiate a rolling submission by year-end and complete the BLA by the end of the first quarter of 2025, enabling potential approval in the U.S. by the end of 2025 if priority review were granted. Following this, in a press release on December 18, 2024, Pauls stated that the Company expected to complete the rolling BLA by the end of the first quarter of 2025. By March 6, 2025, the message was that completion was near and still on track for the end of the first quarter.
Investors allege that, behind these assurances, the MOLBREEVI BLA lacked sufficient CMC information, making FDA approval in its then-current form unlikely and rendering the promised submission timeline unrealistic. The complaint further alleges that the regulatory delay increased the likelihood that the Company would need to raise additional capital, and that the public statements were materially false and misleading throughout the Class Period.
The Truth Emerges
The alleged truth surfaced on May 27, 2025, when Savara announced it had received an FDA Refusal to File letter for the MOLBREEVI BLA. The Company disclosed that, upon preliminary review, the FDA determined the BLA submitted in March 2025 was not sufficiently complete to permit substantive review and requested additional CMC data.
According to the complaint, this disclosure called into question prior statements that the rolling submission would be completed by the end of the first quarter of 2025 and that a path to potential approval by year-end 2025, if priority review were granted, was open. On August 13, 2025, after the Class Period, Savara issued a press release that it now planned to resubmit the MOLBREEVI BLA in December 2025.
Market Reaction
The market reacted the same day. On May 27, 2025, following the disclosure of the FDA's Refusal to File letter, Savara's stock fell $0.90 per share, or 31.69%, to close at $1.94 per share. The complaint ties this decline to the revelation that the BLA was not sufficiently complete and required additional CMC information.
Next Steps
- Submissions for lead plaintiff are due November 7, 2025.
- The Court will issue its order for lead plaintiff and counsel in the weeks after submissions are due.
- The Court will then consider motion for class certification.
- The Court will later consider a Motion to Dismiss.
To learn if you are eligible for recovery under the SVRA securities class action lawsuit, visit the case submission page here.
Disclaimer: This shareholder alert is for informational purposes only and does not constitute legal advice. Consult a qualified attorney for personalized guidance. No specific outcomes are guaranteed.
![Savara Inc. (SVRA) Securities Class Action Lawsuit Filed [October 13, 2025]](https://dropinblog.net/cdn-cgi/image/fit=scale-down,format=auto,width=700/34260815/files/featured/svra-savara-inc-securities-lawsuit-blog-banner.webp)


![V.F. Corporation (VFC) Securities Class Action Lawsuit Filed [October 13, 2025]](https://dropinblog.net/cdn-cgi/image/fit=scale-down,format=auto,width=700/34260815/files/featured/vfc-banner-image.webp)
![Lantheus Holdings, Inc. (LNTH) Securities Class Action Lawsuit Update [October 9, 2025]](https://dropinblog.net/cdn-cgi/image/fit=scale-down,format=auto,width=700/34260815/files/featured/lantheus-securities-class-action-lawsuit-blog-banner.webp)