Gossamer Bio, Inc. Class Action Lawsuit - GOSS
|
Gossamer Bio Class Action Summary |
|
|
Company |
Gossamer Bio, Inc. (NASDAQ: GOSS) |
|
Lead Plaintiff Deadline |
June 1, 2026 |
|
Class Period |
June 16, 2025 – February 20, 2026 |
|
Stock Drop |
February 23, 2026 – GOSS fell $1.71 (over 80%) to $0.42 |
|
Lawsuit Type |
Securities Class Action |
Introduction
A securities class action lawsuit has been filed against Gossamer Bio, Inc. (NASDAQ: GOSS) and its CEO Faheem Hasnain on behalf of investors who purchased or acquired Gossamer Bio securities between June 16, 2025, and February 20, 2026. The complaint, filed by Levi & Korsinsky, LLP on behalf of plaintiff Daniel Kinnamon in the United States District Court for the Southern District of California, alleges that defendants made materially false and misleading statements regarding the design and prospects of the Company's Phase 3 PROSERA study evaluating seralutinib for the treatment of pulmonary arterial hypertension (PAH). Specifically, the lawsuit claims defendants failed to disclose critical trial design issues at the study's Latin American clinical sites, where a heavily-treated, lower-risk patient population performed unusually well on placebo, ultimately causing the study to miss its primary endpoint. When Gossamer Bio disclosed topline results on February 23, 2026, the stock price collapsed from $2.13 to $0.42 per share, a decline of over 80% in a single trading day.
Company Profile
Gossamer Bio, Inc. is a clinical-stage biopharmaceutical company focused on the development and commercialization of seralutinib for the treatment of pulmonary hypertension associated with interstitial lung disease. The Company is headquartered in San Diego, California, and its common stock trades on the NASDAQ under the ticker symbol GOSS.
Class Period
June 16, 2025 – February 20, 2026, inclusive.
Investors who purchased or acquired Gossamer Bio, Inc. (GOSS) securities during the Class Period may be entitled to seek recovery under the federal securities laws.
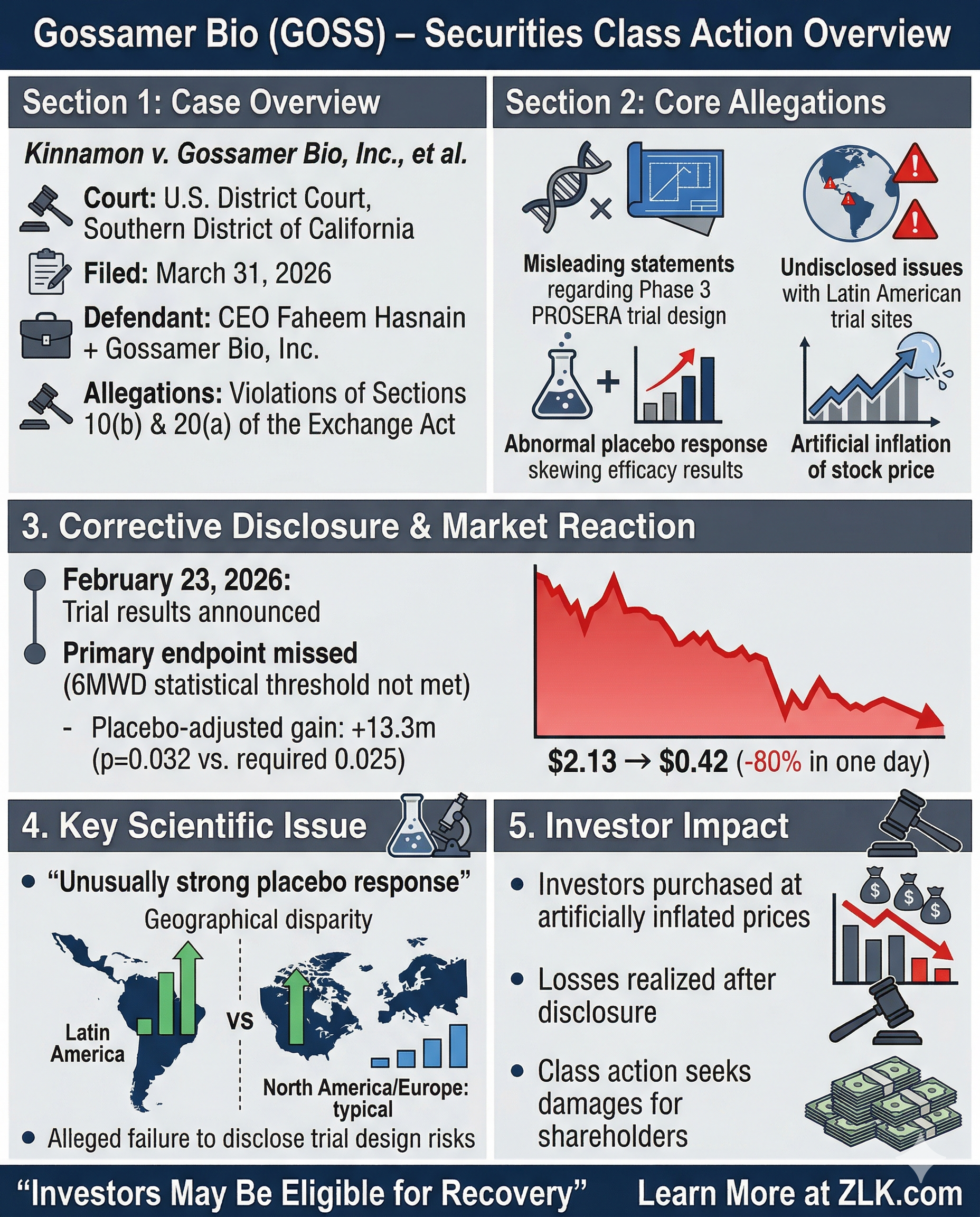
Allegations
The complaint alleges that throughout the Class Period, Gossamer Bio and its CEO Faheem Hasnain projected confidence in the Phase 3 PROSERA study, a registrational trial evaluating seralutinib's ability to improve six-minute walk distance (6MWD) in PAH patients at Week 24. Beginning with a June 16, 2025 press release announcing the completion of PROSERA enrollment, Hasnain told investors the Company had "focused on selecting a patient population that aligns closely with the study's objectives," stating that based on preliminary baseline characteristics, "we firmly believe that we have accomplished this patient selection goal." This set the tone for a series of statements that the complaint alleges painted an overwhelmingly positive picture of PROSERA's trajectory while concealing known deficiencies in the trial's design.
On August 5, 2025, Hasnain reinforced this narrative, describing the upcoming PROSERA readout as "the foundation" of a potential multi-billion-dollar franchise and expressing "conviction around the strength of the science." He told investors the team was "executing the PROSERA Study with discipline and operational excellence" and looked forward to sharing topline results in February. On November 5, 2025, Hasnain again highlighted the Company's progress, stating Gossamer was "progressing through the final stages of the PROSERA Phase 3 Study" and calling it "a pivotal moment."
According to the complaint, these statements were materially false and misleading because defendants knew or recklessly disregarded that patients enrolled at the Latin American clinical sites were largely heavily-treated and lower-risk, a population that would perform particularly well on placebo. As the drug's sponsor, defendants had access to non-public information about the trial's design, clinical site selection, and the protocol issues that posed acute risks to the study meeting its primary endpoint. The complaint alleges defendants constructed a narrative that PROSERA would succeed while failing to disclose the specific vulnerabilities in the Latin American testing sites that ultimately undermined the trial's statistical outcome.
The Truth Emerges
On February 23, 2026, Gossamer Bio published a press release and hosted a Special Call revealing that the Phase 3 PROSERA study had failed to meet its primary endpoint. While seralutinib showed a +28.2 meter improvement in 6MWD from baseline compared to +13.5 meters for placebo, the placebo-adjusted gain of +13.3 meters carried a p-value of 0.0320, failing to clear the prespecified 0.025 alpha threshold required for statistical significance. CEO Hasnain acknowledged the Company had "narrowly missed the stringent prespecified statistical threshold," while Chief Medical Officer Richard Aranda disclosed that "the placebo arm showed a larger improvement that is often seen in many other Phase III PH trials" and that "in other regions, particularly Latin America, outsized placebo improvements materially compressed the pool treatment difference."
During the question-and-answer session, COO and CFO Bryan Giraudo provided further detail that directly contradicted the Company's prior optimism. He disclosed that Gossamer had made "a significant investment in Latin America" expecting it to be the study's best-performing geography based on results from the STELLAR study for sotatercept, but instead found "an almost parity between the placebo rate and the treatment rate" in the region, a result he called "extremely, extremely disturbing." He acknowledged the Company was "still early in the investigation of what happened in Latin America." These revelations stood in stark contrast to defendants' prior statements, which the complaint alleges never mentioned any trial design concerns regarding the Latin American sites. Analyst reports following the disclosure confirmed the significance: Oppenheimer characterized the Latin American anomaly as "a trial execution issue, not a drug effect issue" and suggested the irregularity pointed to "human measurement error during 6MWD lap counting," while Wedbush downgraded Gossamer to Neutral and slashed its price target from $6 to $1.
Market Reaction
The market response to PROSERA's failure was immediate and severe. On February 23, 2026, Gossamer Bio's stock price plummeted from a closing price of $2.13 per share on February 20, 2026, to just $0.42 per share, a decline of over 80% in a single trading day. The over 80% decline reflected the market's reaction to learning that the Phase 3 PROSERA study had missed its primary endpoint due to trial design defects in Latin America that defendants had never disclosed during the Class Period.
Next Steps
● The Court will issue its order for lead plaintiff and counsel in the weeks after submissions are due.
● The Court will then consider motion for class certification.
● The Court will later consider a Motion to Dismiss.
Disclaimer: This shareholder alert is for informational purposes only and does not constitute legal advice. Consult a qualified attorney for personalized guidance. No specific outcomes are guaranteed.
