Introduction to Alto Neuroscience, Inc. (ANRO) Securities Lawsuit
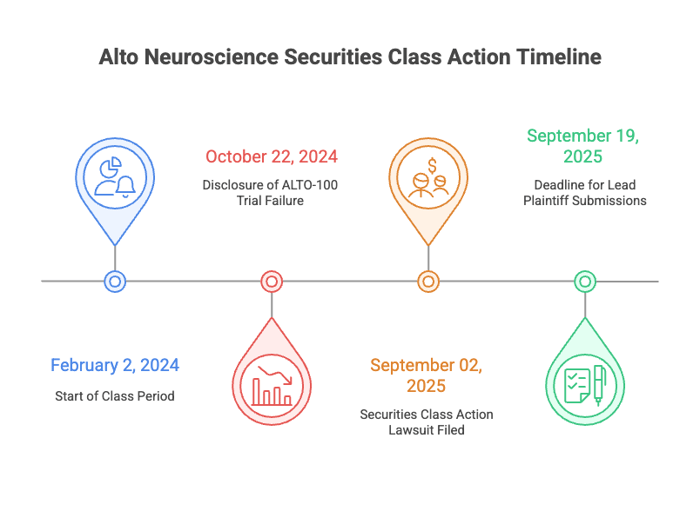
Investors hit Alto Neuroscience, Inc. (NYSE: ANRO) with a securities class action suit. The lawsuit represents investors who got Alto stock between February 2, 2024, and October 22, 2024 (the “Class Period”). Plaintiffs say Alto and some of its execs released materially false and misleading information about one of its major drugs, ALTO-100 (for treating major depressive disorder).
According to the complaint, Alto led investors to think ALTO-100 as more promising than it was and touted its strong regulatory and commercial prospects. In reality, the drug was less effective, leading to trial failure disclosure. Shareholders are now seeking to recover their losses.
Alto Neuroscience, Inc. (ANRO) Securities Lawsuit Case Details
Caption: Feldman v. Alto Neuroscience, Inc., et al.
Case No.: 5:25-cv-06105-NW
Jurisdiction: U.S. District Court, Northern District of California
Filed on: July 21, 2025
Alto Neuroscience, Inc. (ANRO) Company Profile
Alto is a clinical-stage pharma company. It makes unique therapies for psychiatric disorders using its precision psychiatry platform. ALTO-100 is one of Alto’s major drug candidates. ALTO-100 was in Phase 2b trials for major depressive disorder at the time of the Alto’s IPO.
Class Period:
February 2, 2024 through October 22, 2024
Investors who purchased Alto securities during the class period might be eligible to join the Alto Neuroscience securities class action lawsuit.

Allegations in the Alto Neuroscience, Inc. (ANRO) Securities Fraud
Alto and its execs fed investors a line about ALTO-100's prospects. They hid inconvenient facts from IPO offering documents, press releases, and SEC filings. These omissions made the drug look more effective than it was; that helped prop up stock prices . . . until the whole web of misstatements came crumbling down.
It started with the IPO documents touting ALTO-100. The February 2, 2024 IPO paperwork said ALTO-100 said it is a unique, scientifically-complex drug with first-in-class potential. Alto execs highlighted great Phase 2a results where cognitively impaired MDD patients responded better. The company pushed the drug into to Phase 2b in 266 biomarker-characterized patients.
Financial reports backed the hype. The FY23 Form 10-K on March 21, 2024, repeated the praise, with CEO Etkin calling 2024 transformational. The company touted that ALTO-100 data would be ready in second half of 2024.
May's Q1 press release kept up the spin. CEO Etkin said the Alto team was committed to execution with five Phase 2 studies initiated and data over next 12 months. By July 16, the Phase 2b enrollment was completed. Alto’s CEO bragged that this was significant achievement; meanwhile, Alto’s Chief Medical Officer emphasized the potential for targeted therapies. A July 25, 2024 announcement of a funding award for bipolar depression included statements from the CEO regarding the opportunity to address a subset of patients with similar profiles. In its August 2024 quarterly release, Alto stated it had completed its first biomarker-based study and reiterated that topline data would be available in October.
The complaint asserts that these statements were materially misleading because defendants allegedly knew, or were reckless in not knowing, that ALTO-100 was less effective than portrayed. On October 22, 2024 Alto admitted the Phase 2b study didn’t meet primary endpoint. That admission was a stark change from the all the big talk about ALTO-100 which lead up to that point.
Plaintiff says Alto’s misstatements and omissions artificially inflated the company’s stock price during the Class Period. When the truth emerged, the stock took a hit and investors lost millions.
The Truth Emerges
On October 22, 2024, the Company revealed the Phase 2b study of ALTO-100 in MDD did not meet its primary endpoint. Despite all the pre-study hype, there was no change on critical measures versus placebo.
Market Reaction
The stock took a hit: After the October 2024 news, it plunged 70%.
The stock lost a jaw-dropping 80% -- or $17.44 per share -- between its high in February 2024 and its collapse in October 2024. Over $600 million was wiped out from that freefall.
Next Steps
- Submissions for lead plaintiff are due September 19, 2025.
- The Court will issue its order for lead plaintiff and counsel in the weeks after submissions are due.
- The Court will then consider motion for class certification.
- The Court will later consider a Motion to Dismiss.
To learn if you are eligible for recovery under the Alto securities class action lawsuit, visit the case submission page here.
Disclaimer: This shareholder alert is for informational purposes only and does not constitute legal advice. Consult a qualified attorney for personalized guidance. No specific outcomes are guaranteed.
![Alto Neuroscience, Inc. (ANRO) Securities Class Action Lawsuit [September 02, 2025]](https://dropinblog.net/cdn-cgi/image/fit=scale-down,format=auto,width=700/34260815/files/featured/altoneuroscience-securities-lawsuit-blog-banner.webp)



![RxSight, Inc. (RXST) Securities Class Action Lawsuit Update [September 03, 2025]](https://dropinblog.net/cdn-cgi/image/fit=scale-down,format=auto,width=700/34260815/files/featured/rxsight-securities-lawsuit-blog-banner.webp)