A federal court has dismissed a securities fraud lawsuit against Anavex Life Sciences Corp. (NASDAQ: AVXL), ending the case with prejudice and preventing shareholders from refiling claims.
What Was the Case About?
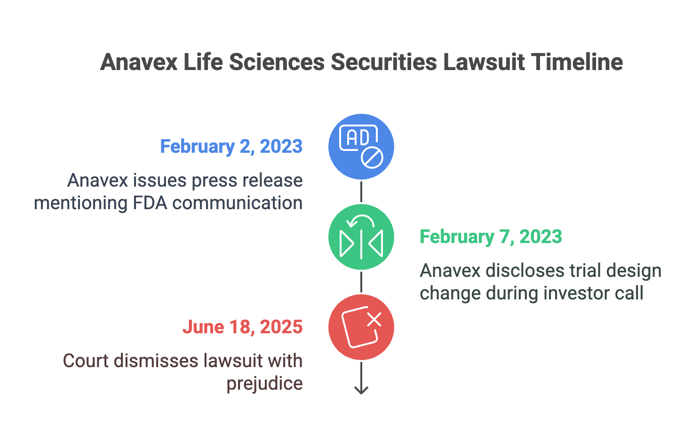
The lawsuit, filed in the U.S. District Court for the Southern District of New York, accused Anavex and its CEO, Christopher U. Missling, of misleading investors about the company's Rett syndrome drug trial. Shareholders claimed that Anavex repeatedly told the market it would use the same primary endpoint (RSBQ AUC) in its pediatric EXCELLENCE trial that it used the previous AVATAR trial. AVATAR was also a study about the same Rett syndrome drug.
The complaint said that Anavex got feedback from regulators urging it to change that endpoint. Despite getting that critical feedback, Anavex allegedly left shareholders in the dark. The company eventually did come clean and admit the the change during a February 7, 2023 investor call. However, shareholders argued that the revelation harmed the stock’s value and caused investor losses.
Anavex Responds with Motion to Dismiss
Anavex pushed back hard. In its motion to dismiss, the company argued that the allegedly “false or misleading” statements at issue were either true when made, forward-looking, or too vague to form the basis of securities fraud. The company also said there was no proof of intent to deceive investors. Finally, Anavex highlighted that its stock price actually rose by nearly 6% on the day of the disclosure, undermining any claim of investor losses.
Plaintiff’s Argument
The plaintiff said Anavex had a legal obligation to update the market once it received FDA feedback requiring a methodology change. The plaintiff also argued a February 2 press release mentioning FDA communication was misleading because it failed to explain the change in trial design. Even though the stock rose on February 7, they claimed declines on February 8 and 9 showed a delayed market response, which should satisfy the loss causation standard at the pleading stage.
Court Sides with Anavex
On June 18, 2025, Judge Colleen McMahon granted Anavex’s motion to dismiss with prejudice. She found that only one of the ten alleged misstatements (the February 2 press release) was plausibly misleading. But even that single claim failed, because the plaintiff couldn’t show loss causation. The court noted that Anavex’s stock rose on February 7, 2023, the day of the supposed corrective disclosure, and cited binding Second Circuit case law to dismiss the claim.
Final Decision
The case is now closed. The court’s decision to dismiss with prejudice means the claims cannot be refiled. No further amendments to the complaint will be allowed.
What This Means for Shareholders
If you're an Anavex Life Sciences shareholder and have questions about this decision or your legal options, you can contact Levi & Korsinsky at www.zlk.com.
This alert is for informational purposes only and does not constitute legal advice.




