Introduction to Capricor Therapeutics, Inc. (CAPR) Securities Lawsuit
Capricor Therapeutics, Inc. (NASDAQ: CAPR), a biotech company, got slapped with a securities fraud suit. The complaint is for people who got Capricor stock between October 9, 2024, and July 10, 2025 (the “Class Period”). The case claims that Capricor and certain executives misled the public about deramiocel for DMD cardiomyopathy, concealing adverse facts about four-year safety and efficacy data from Phase 2 HOPE-2 trial.
According to the complaint, Capricor portrayed its BLA submission as strong. In reality, the data did not support effectiveness, leading to FDA denial. Shareholders are now seeking to recover their losses.
Capricor Therapeutics, Inc. (CAPR) Securities Lawsuit Case Details
Citation: Hau Xiang Leong v. Capricor Therapeutics, Inc., et al.
Case No.: 3:25-cv-01815-DMS-AHG
Jurisdiction: U.S. District Court, Southern District of California
Filed on: July 17, 2025
Capricor Therapeutics, Inc. (CAPR) Company Profile
Capricor Therapeutics, Inc. (NASDAQ: CAPR is a biotech company. Capricor’s business focuses on developing cell and exosome-based therapies for rare diseases. Its lead candidate, deramiocel, targets cardiomyopathy associated with Duchenne muscular dystrophy (DMD).
Class Period:
October 9, 2024, to July 10, 2025
Investors who purchased Capricor securities during the class period might be eligible to join the Capricor class action lawsuit.

Allegations in the Capricor Therapeutics, Inc. (CAPR) Securities Lawsuit
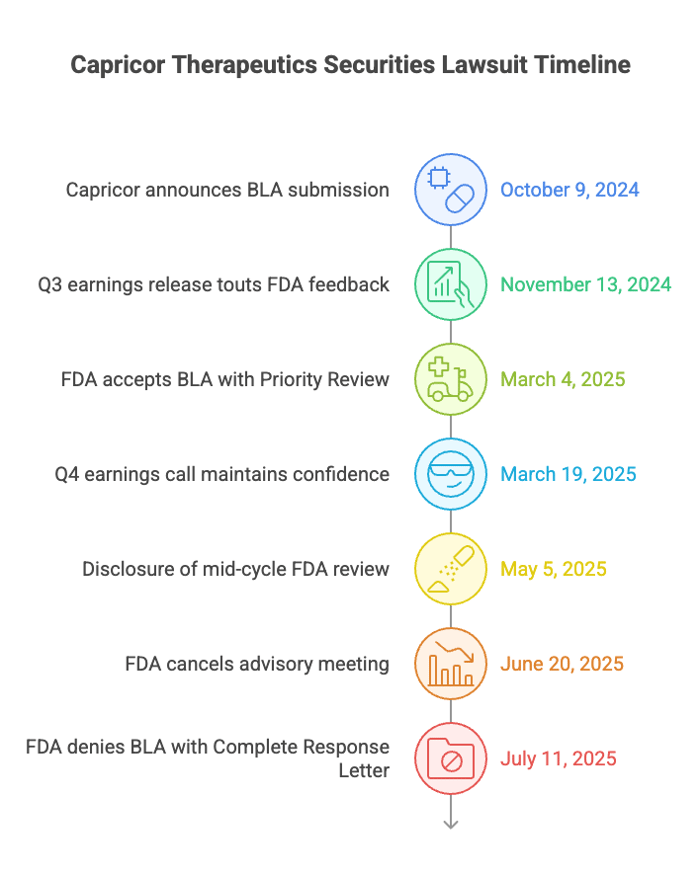
From October 9, 2024 to July 10, 2025, Capricor Therapeutics kept touting its chances of getting the FDA’s nod of approval on a BLA for deramicoel. For instance, on October 9, 2024, Capricor announced the roll-out of the BLA submission. It even claimed the drug was likely eligible priority review. In its Q3 earnings release on November 13, 2024, the company emphasized FDA feedback and claimed statistically significant efficacy from HOPE-2 and HOPE-2 OLE trials. CEO Linda Marbán stated the BLA would be complete by year-end and touted commercialization readiness.
On March 4, 2025, Capricor said FDA accepted the BLA. The FDA gave it a Priority Review designation and a PDUFA date of August 31, 2025. On March 19, 2025 Q4 earnings call, execs kept up the confidence in the drug and downplayed the need for an advisory committee. On May 5, 2025, Capricor confirmed the FDA would hold an advisory meeting, yet continued to assert no major issues had been identified.
The complaint alleges these statements concealed material adverse facts about the four-year HOPE-2 data, which failed to meet statistical thresholds under the original model. The FDA ultimately denied the BLA on July 11, 2025, citing lack of substantial evidence of effectiveness and unresolved Chemistry, Manufacturing, and Controls (CMC) issues. Investors suffered steep losses as the truth emerged.
The Truth Emerges
The illusion of regulatory momentum unraveled in stages.
On May 5, 2025, Capricor disclosed a mid-cycle FDA review with no identified deficiencies and an advisory committee meeting planned—yet the stock still fell.
On June 20, 2025, Stat News reported the FDA cancelled the meeting. The government cited skepticism about deramiocel’s safety and efficacy as reasons for pulling the plug on the meeting. Investors weren’t pleased: Capricor’s stock price plummeted by 31% in two days.
Things got worse for Capricor. On July 11, 2025, the FDA issued a Complete Response Letter (CRL) denying the BLA. The agency said there wasn’t enough evidence the drug actually worked. They also cited ongoing issues with Chemistry, Manufacturing, and Controls. Capricor’s prior assurances stood in contrast to the FDA’s final position. Each disclosure stripped away investor certainty, culminating in a 33% single-day drop. The data, long touted as compelling, failed to meet the threshold for approval.
Market Reaction
Stock declined sharply on disclosures: 29% on May 5, 2025; 31% over June 18-20, 2025; 33% on July 11, 2025.
Next Steps
- Submissions for lead plaintiff are due September 15, 2025.
- The Court will issue its order for lead plaintiff and counsel in the weeks after submissions are due.
- The Court will then consider motion for class certification.
- The Court will later consider a Motion to Dismiss.
To learn if you are eligible for recovery under the Capricor securities class action lawsuit, visit the case submission page here.
Disclaimer: This shareholder alert is for informational purposes only and does not constitute legal advice. Consult a qualified attorney for personalized guidance. No specific outcomes are guaranteed.
![Capricor Therapeutics, Inc. (CAPR) Securities Class Action Lawsuit Update [August 25, 2025]](https://dropinblog.net/cdn-cgi/image/fit=scale-down,format=auto,width=700/34260815/files/featured/capricor-therapeutics-securities-lawsuit-blog-banner.webp)


![Neogen Corporation (NEOG) Securities Class Action Lawsuit Update [August 26, 2025]](https://dropinblog.net/cdn-cgi/image/fit=scale-down,format=auto,width=700/34260815/files/featured/neogen-securities-lawsuit-blog-banner.webp)
