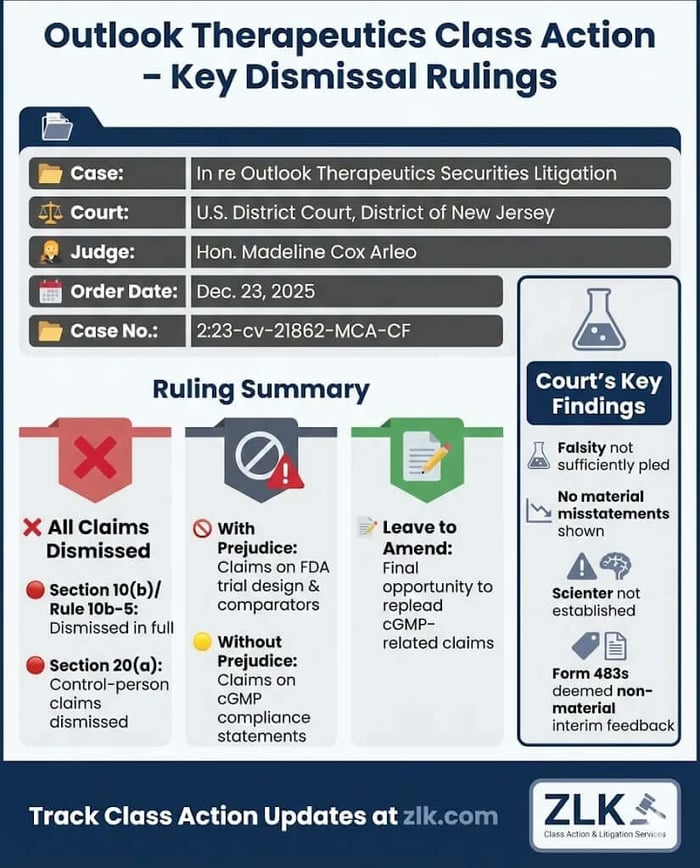
Caption: In re Outlook Therapeutics, Inc. Securities Litigation.
Case No.: 2:23-cv-21862-MCA-CF
Jurisdiction: U.S. District Court, District of New Jersey
Judge: Hon. Madeline Cox Arleo

Summary
On December 23, 2025, Judge Madeline Cox Arleo granted defendants’ motion to dismiss the Consolidated Second Amended Complaint in its entirety. The Court dismissed all Exchange Act claims under Section 10(b) and Rule 10b-5, and the derivative Section 20(a) claims. Certain claims were dismissed with prejudice, while others were dismissed without prejudice, with plaintiffs given one final opportunity to amend.
Allegations Against Outlook Therapeutics, Inc.
Plaintiffs alleged that Outlook Therapeutics and two senior executives made materially false or misleading statements during the class period concerning the likelihood of FDA approval for ONS-5010. The complaint alleged that defendants concealed information about FDA communications, clinical trial design, and manufacturing compliance issues at third-party facilities. Plaintiffs claimed these omissions misled investors about regulatory risk and approval prospects.
Defendants’ Motion to Dismiss
Outlook and the individual defendants moved to dismiss on the grounds that plaintiffs failed to plead falsity, scienter, and materiality with the particularity required by Rule 9(b) and the PSLRA. They argued that the challenged statements were accurate, non-actionable opinion or puffery, or unsupported by contemporaneous facts showing defendants knew approval was unlikely. Defendants also argued that Form 483 observations issued to third-party manufacturers were not material and were not shown to be known to defendants.
Plaintiffs’ Opposition
Plaintiffs argued that FDA guidance, the size of prior approved trials, Form 483s issued to manufacturing partners, and confidential witness allegations supported an inference that defendants knew FDA approval was unlikely. They contended that defendants’ statements created a misleading impression about trial sufficiency, regulatory compliance, and manufacturing readiness.
Court’s Ruling
The Court dismissed all Section 10(b) and Rule 10b-5 claims. Claims based on alleged misstatements about FDA trial design and comparator trials were dismissed with prejudice. Claims based on alleged misstatements regarding cGMP compliance were dismissed without prejudice. The Court dismissed the Section 20(a) control-person claims in their entirety.
Court’s Rationale
Falsity: The Court held that plaintiffs failed to plead facts showing that statements about FDA interactions, trial design, or manufacturing compliance were false or misleading when made. Many statements did not claim FDA agreement on approval standards, and others accurately described interactions or expressed opinion.
Materiality: The Court found that Form 483 observations, standing alone, were interim regulatory feedback and not material absent allegations of follow-up FDA action or known violations.
Scienter: Plaintiffs failed to plead contemporaneous facts showing defendants knew approval was unlikely or that challenged statements were misleading.
Section 20(a): Because plaintiffs failed to plead a primary violation, the control-person claims necessarily failed.
Leave to Amend: Amendment was deemed futile for claims based on FDA trial design and comparator statements but permitted one final attempt to replead claims based on cGMP compliance.
Case Status
All claims have been dismissed. Claims based on FDA trial design and comparator statements are dismissed with prejudice. Claims based on cGMP compliance statements are dismissed without prejudice, and plaintiffs may file a final amended complaint.




