Table of Contents
Introduction to Hims & Hers Health, Inc. (HIMS) Securities Lawsuit
Hims & Hers Health, Inc. (NYSE: HIMS) got slapped with a securities fraud class action lawsuit. It’s for investors who bought stock between April 29, 2025, and June 22, 2025 (the “Class Period”). The lawsuit claims Hims lied about its Novo Nordisk deal and peddled dodgy weight loss drugs. They hyped FDA-approved Wegovy® like a miracle pill—then got caught pushing knockoffs. Stocks tanked. Investors lost millions. Shareholders are out for blood.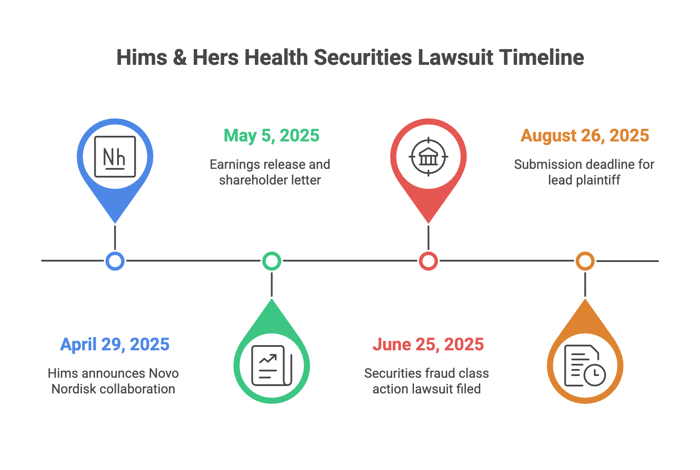
Hims & Hers Health, Inc. (HIMS) Lawsuit Case Details
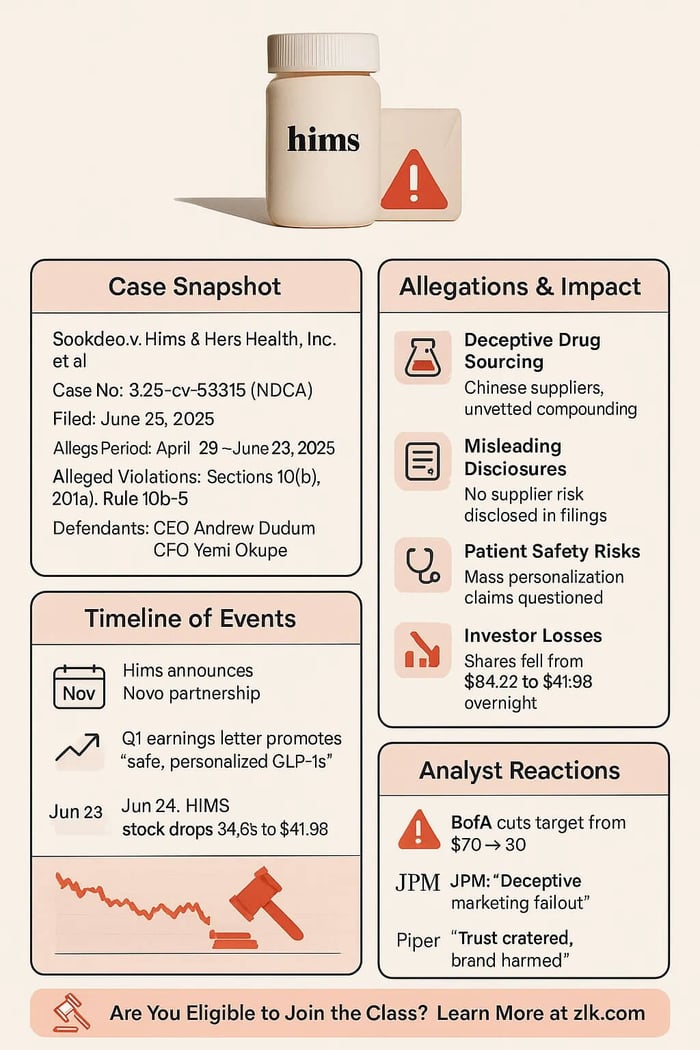
Sookdeo v. Hims & Hers Health, Inc., et al.
Case No.: 3:25-cv-05315
U.S. District Court, Northern District of California
Filed on June 25, 2025
Hims & Hers Health, Inc. (HIMS) Company Profile
Hims (NYSE: HIMS) runs a telehealth platform. It connects folks to doctors to treat a wide-range of ailments, like sexual health, mental health, skin care, weight loss. It operates in the U.S. and overseas. Hims sells its products via apps, websites, and wholesale partners.
Class Period: April 29, 2025 – June 22, 2025
Investors who purchased HIMS securities during the Class Period might be eligible to join the Hims class action lawsuit.
Allegations in the Hims & Hers Health, Inc. (HIMS) Securities Lawsuit
Hims’ telehealth model delivers quick access to medications, including weight loss drugs like Wegovy (semaglutide for obesity. Its partnership with Novo Nordisk, Wegovy®’s maker, promised legitimate supply. But FDA rules tightly control compounding. Hims’ growth relied on these compounded drugs, yet it concealed sourcing from unapproved suppliers. That set up up a future regulatory disaster.
Plaintiffs say Hims and its executives violated securities law under the Securities Exchange Act. On April 29, 2025, Hims issued a press release announcing a “long-term collaboration” with Novo Nordisk. Hims was going to distribute “FDA-approved Wegovy®” with clinical support through its telehealth platform, in collaboration with Novo.
On May 5, 2025, Hims’ earnings release and shareholder letter touted “personalized solutions” like compounded GLP-1s and a generic tiraglutide launch, plus “innovative partnerships” driving growth. The Q1 2025 Form 10-Q highlighted increased platform traffic but omitted risks tied to unapproved compounding or regulatory exposure.
What Hims failed to tell investors was Hims marketed compounded semaglutide from unapproved Chinese suppliers, not Novo Nordisk’s authorized chain. The complaint says these omissions inflated Hims’ stock price, hiding a looming regulatory collapse.
This lawsuit accuses Hims with violating federal securities laws, including Sections 10(b) and 20(a) of the Securities Exchange Act. Plaintiffs’ legal claims state that Hims’ statements about its Wegovy® offerings and partnerships were materially false and misleading, lacking a reasonable basis, and harmed investors.
The Truth Emerges
On June 23, 2025, Novo Nordisk dropped the hammer with press release skewering Hims. Novo blasted Hims’ “illegal mass compounding” and “deceptive marketing that put patient safety at risk.”
Novo axed the partnership. Analysts screamed litigation risk and brand damage, with Hims’ drug push exposed as a sham.
Market Reaction
On June 24, 2025, Hims stock fell 34.6%, closing at $41.98, down from $64.08 the prior day. The drop followed Novo Nordisk’s disclosure and widespread media coverage.
Next Steps
- Submissions for lead plaintiff are due August 26, 2025.
The Court will issue its order for lead plaintiff and counsel in the weeks after submissions are due.
The Court will then consider motion for class certification.
The Court will later consider a Motion to Dismiss.
To learn if you are eligible for recovery under the Hims securities class action lawsuit, visit the case submission page here.
Disclaimer: This shareholder alert is for informational purposes only and does not constitute legal advice. Consult a qualified attorney for personalized guidance. No specific outcomes are guaranteed.
![Hims & Hers Health, Inc. (HIMS) Securities Class Action Lawsuit Update [July 28, 2025]](https://dropinblog.net/cdn-cgi/image/fit=scale-down,format=auto,width=700/34260815/files/featured/hims-hers-securities-lawsuit-blog-banner-1.webp)

![3D Systems Corp (DDD) Securities Class Action Lawsuit Update [July 20, 2025]](https://dropinblog.net/cdn-cgi/image/fit=scale-down,format=auto,width=700/34260815/files/featured/3d-systems-corporation-ddd-securities-class-action-lawsuit-blog-banner.webp)