Introduction to Sarepta Therapeutics, Inc. (SRPT) Securities Lawsuit
A securities fraud class action lawsuit has been filed against Sarepta Therapeutics, Inc. (NASDAQ: SRPT) on behalf of investors who purchased the Company’s common stock between June 22, 2023, and June 24, 2025 (the “Class Period”). The lawsuit alleges that Sarepta and certain executives violated federal securities laws by making false and misleading statements regarding the safety, efficacy, and regulatory prospects of ELEVIDYS, its gene therapy for Duchenne muscular dystrophy.
Sarepta Therapeutics, Inc. (SRPT) Lawsuit Case Details
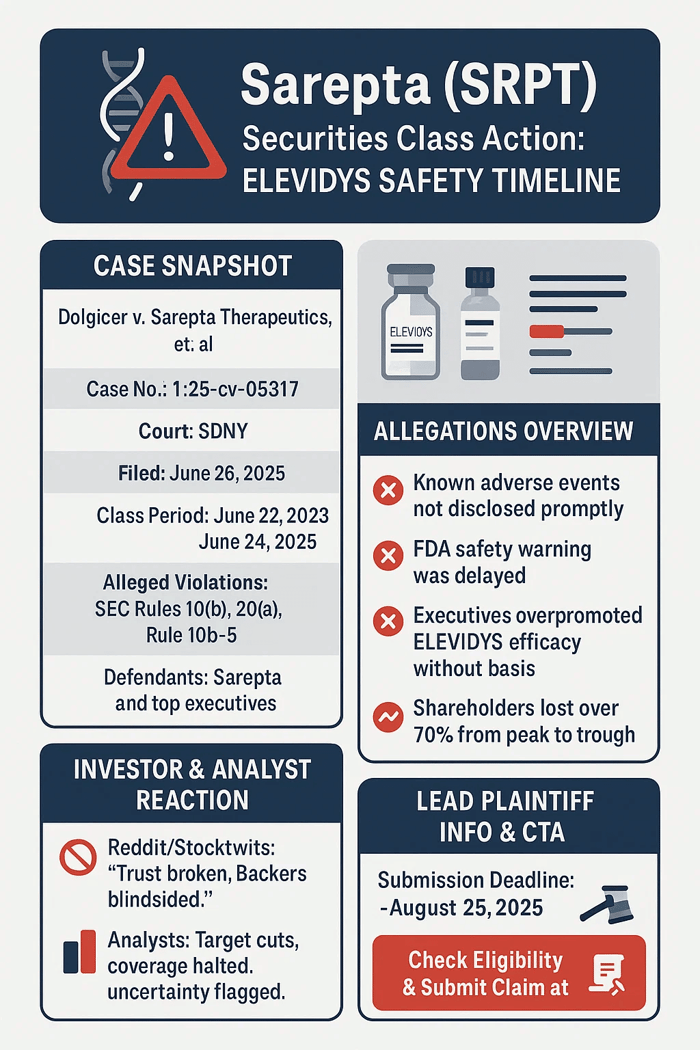
Caption: Dolgicer v. Sarepta Therapeutics, Inc., et al.
Case No: 1:25-cv-05317.
Jurisdiction; U.S. District Court, Southern District of New York.
Filed on June 26, 2025.
Sarepta Therapeutics, Inc. (SRPT) Company Profile
Sarepta is a commercial-stage biopharmaceutical company focused on RNA and gene therapies for rare diseases. It develops treatments for Duchenne muscular dystrophy, including ELEVIDYS, a prescription gene therapy for certain patients. The company trades as SRPT on NASDAQ.
Class Period:
June 22, 2023 – June 24, 2025. Investors who purchased Sarepta securities during the class period might be eligible to join the Sarepta class action lawsuit.
Allegations in the Sarepta Therapeutics, Inc. (SRPT) Securities Lawsuit
The complaint alleges Sarepta and executives Douglas S. Ingram, Dallan Murray, and Louise Rodino-Klapac misled investors about ELEVIDYS. On June 22, 2023, a press release. Sarepta announced FDA approval of ELEVIDYS for ambulatory pediatric patients aged 4-5 with Duchenne. Ingram called it a watershed moment for gene therapy. On August 2, 2023, Q2 earnings claimed a strong ELEVIDYS launch. Ingram said confirmatory trial EMBARK would read out Q4 2023, supporting broader label. On the August 2 call, Ingram said the launch was going well. Rodino-Klapac touted published data showing ELEVIDYS altered disease with no safety issues. Murray noted 700 antibody kits distributed, signaling demand.
On November 1, 2023, Q3 earnings reported $309.3 million revenue, up 49%, with $69.1 million from ELEVIDYS, no new safety signals. Ingram called it a defining moment. On the November 1 call, Ingram said EMBARK stabilized muscle across ages with a laudable safety profile. Murray claimed 70 sites ready to dose. On February 28, 2024, Q4 earnings said FDA accepted a BLA supplement for label expansion. Ingram eyed 2024 as profound. On the February 28 call, Rodino-Klapac said data showed benefits across ages.
On May 2, 2024, Q1 earnings reported $133.9 million ELEVIDYS revenue, $334 million total since launch. Ingram noted significant demand on the May 2 call. On June 20, 2024, a press release announced FDA approval for patients 4+ regardless of ambulatory status. Ingram called it a watershed occasion.
On August 7, 2024, Q2 earnings reiterated expansion. Ingram cited MRI data for muscle health. On the August 7 call, Ingram reported $385.4 million revenue, up 51%. Rodino-Klapac said ELEVIDYS worked across disease states. On October 30, 2024, Q3 earnings and January 27, 2025, Q4 earnings claimed sustained EMBARK benefits. Ingram said ELEVIDYS changed Duchenne’s trajectory. On those calls, Ingram and Rodino-Klapac doubled down on safety and efficacy for 80+ patients.
It didn’t hold. ELEVIDYS carried acute liver failure risks. Trials missed severe side effects. Halts and scrutiny followed, threatening approvals. The filed complaint alleges Sarepta’s failure to disclose these risks violated federal securities laws, including Sections 10(b) and 20(a) of the Securities Exchange Act. Plaintiffs’ legal claims state that Sarepta’s statements about ELEVIDYS’s safety and prospects were materially false and misleading, lacking a reasonable basis, and adversely affected investors.
The Truth Emerges
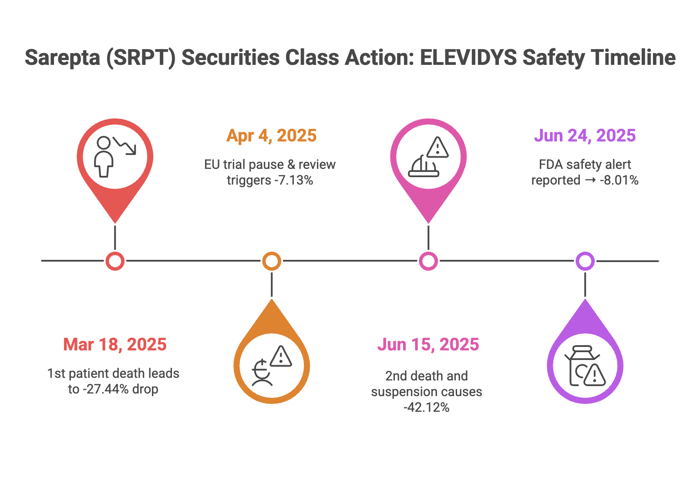
On March 18, 2025, Sarepta announced a patient death from acute liver failure after ELEVIDYS treatment. On April 4, 2025, EU authorities requested data review, halting some trials. On June 15, 2025, a second death led to ELEVIDYS suspension for non-ambulatory patients and a study pause. On June 24, 2025, the Food and Drug Administration (FDA) issued a safety communication investigating acute liver failure risks.
Market Reaction
Sarepta’s stock price fell 27.44% on March 18, 2025, to $73.54. It dropped 7.13% on April 4, 2025, to $54.43. On June 15, 2025, it plunged 42.12% to $20.91. On June 25, 2025, it fell 8.01% to $17.46. These drops cost investors millions.
Next Steps
- Submissions for lead plaintiff are due 60 days after filing.
- The Court will issue its order for lead plaintiff and counsel in the weeks after submissions are due.
- The Court will then consider motion for class certification.
- The Court will later consider a Motion to Dismiss.
To learn if you are eligible for recovery under the Sarepta securities class action lawsuit, visit the case submission page here.
Disclaimer: This shareholder alert is for informational purposes only and does not constitute legal advice. Consult a qualified attorney for personalized guidance. No specific outcomes are guaranteed.

![3D Systems Corp (DDD) Securities Class Action Lawsuit Update [July 20, 2025]](https://dropinblog.net/cdn-cgi/image/fit=scale-down,format=auto,width=700/34260815/files/featured/3d-systems-corporation-ddd-securities-class-action-lawsuit-blog-banner.webp)
![Hims & Hers Health, Inc. (HIMS) Securities Class Action Lawsuit Update [July 28, 2025]](https://dropinblog.net/cdn-cgi/image/fit=scale-down,format=auto,width=700/34260815/files/featured/hims-hers-securities-lawsuit-blog-banner-1.webp)
