Introduction to PepGen Inc. (PEPG) Lawsuit
A securities class action lawsuit has been filed against PepGen Inc. (NASDAQ: PEPG) on behalf of investors who bought PepGen securities between March 7, 2024, and March 3, 2025 (the “Class Period”). The lawsuit claims PepGen overstated the promise of PGN-EDO51, a Duchenne muscular dystrophy (DMD) drug. The lawsuit says PepGen hid weak lab results and safety risks while overhyping the drug’s promise. Investors lost hundreds of millions when the truth broke, prompting the PepGen lawsuit.
PepGen Inc. (PEPG) Securities Lawsuit Case Details
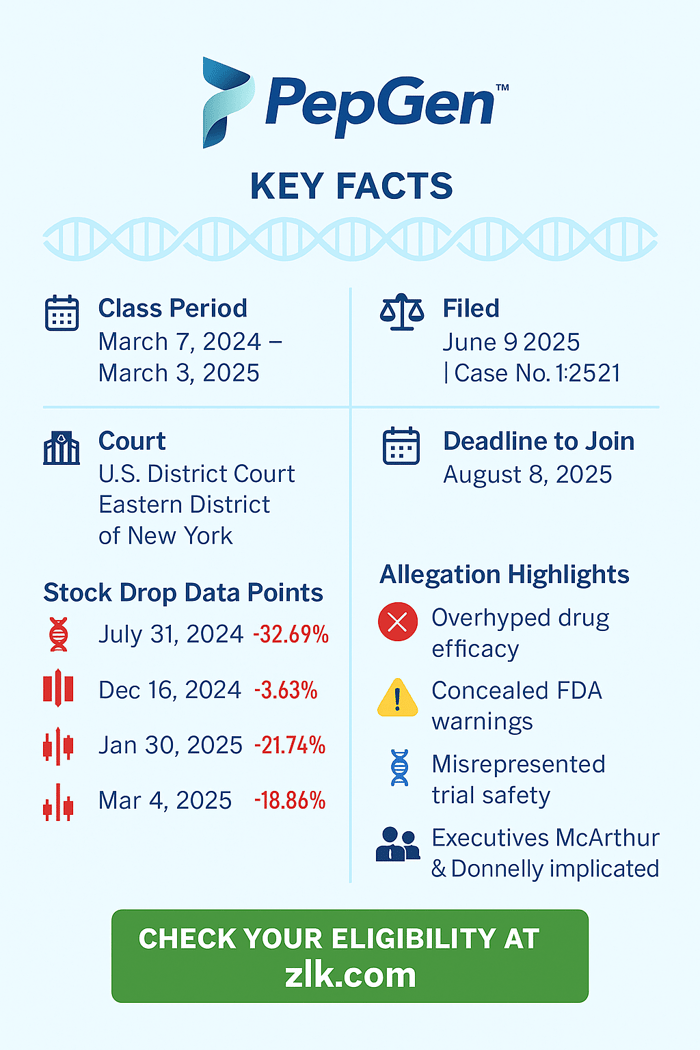
Case Name: Karam v. PepGen Inc., et al.
Case No.: 1:25-cv-03221
U.S. District Court, Eastern District of New York
Filed on June 9, 2025
PepGen Inc. (PEPG) Company Profile
PepGen is Boston-based biotech. It develops oligonucleotide drugs for severe neuromuscular diseases like DMD. DMD is a genetic disorder which causes muscle weakness. PepGen’s leading drug is PGN-EDO51, which seeks to treat DMD.
PepGen conducted two Phase 2 trials on PGN-ENDO51: CONNECT1 and CONNECT2.
Class Period:
March 7, 2024 to March 3, 2025, inclusive
Investors who purchased PepGen securities during the class period might be eligible to join the PepGen securities class action lawsuit.
Allegations in the PepGen Inc. (PEPG) Lawsuit
The complaint alleges PepGen and its executives (James McArthur and Noel Donnelly) violated federal securities law, such as the Securities Exchange Act, by making materially false and misleading claims about the commercial prospects of PNG-EDO51 and its chances for approval by the Food and Drug Administration (FDA).
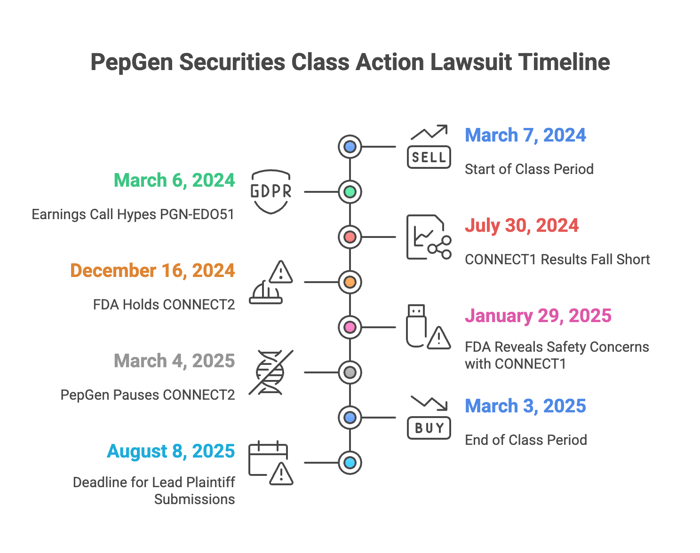
On March 6, 2024, PepGen hosted an earnings call. Executives hyped PGN-EDO51, predicting promising lab results in the CONNECT1 study. Specifically, McArthur predicted over 7-9% dystrophin production in the study’s 10 mg/kg cohort, which is a key success indicator. The Company’s May 14, 2024 press release and Q1 2024 Form 10-Q claimed CONNECT2’s design supported FDA approval. The November 2024 earnings call and Q3 2024 Form 10-Q boasted “favorable safety” and other promising science, projecting a path to market.
But plaintiffs claim the science wasn’t there and the Company’s executives knew it. PGN-EDO51 produced dystrophin levels that were a mere fraction of those asserts. Moreover, the drug posed major safety issues. CONNECT2 was under FDA fire for design deficiencies. But, according to plaintiffs, PepGen hid these issues, propping up the stock price and harming investors.
The filed complaint alleges PepGen’s failure to disclose these known risks violated federal securities laws, including Sections 10(b) and 20(a) of the Securities Exchange Act. Plaintiffs’ legal claims state that PepGen’s false and misleading statements about PGN-EDO51’s efficacy and trial prospects were lacked a reasonable basis and adversely affected investors.
The Truth Emerges
PepGen’s July 30, 2024 press release reported CONNECT1’s results. They fell short of investor expectations. The 5 mg/kg cohort hit only 0.61% dystrophin, which was far below the expected mark. Analysts called it a miss. On December 16, 2024, the FDA put a hold on CONNECT2, citing risks. On January 29, 2025, the FDA revealed safety concerns with CONNECT1. PepGen paused CONNECT2 on March 4, 2025 and scrapped its DMD programs by May 28, 2025.
Market Reaction
Investors were furious. PepGen’s stock fell:
32.69% ($5.55) to $11.43 on July 31, 2024;
3.63% ($0.17) to $4.51 on December 16, 2024;
21.74% ($0.40) to $1.44 on January 30, 2025; and,
18.86% ($0.53) to $2.28 on March 4, 2025,
Collectively costing shareholders hundreds of millions. Investors paid for PepGen’s silence.
Next Steps
Submissions to serve as lead plaintiff are due August 8, 2025.
The Court will issue its order for lead plaintiff and counsel in the weeks after submissions are due.
The Court will then consider motion for class certification.
The Court will later consider a Motion to Dismiss.
To learn if you are eligible for recovery under the PepGen securities fraud lawsuit, visit the case submission page here.
Disclaimer: This shareholder alert is for informational purposes only and does not constitute legal advice. Consult a qualified attorney for personalized guidance. No specific outcomes are guaranteed.
![PepGen Inc. (PEPG) Securities Class Action Lawsuit Update [July 14, 2025]](https://dropinblog.net/cdn-cgi/image/fit=scale-down,format=auto,width=700/34260815/files/featured/pepgen-lawsuit-banner.webp)

![Vestis Corporation (VSTS) Securities Class Action Lawsuit Update [July 12, 2025]](https://dropinblog.net/cdn-cgi/image/fit=scale-down,format=auto,width=700/34260815/files/featured/vestis-corp-securities-lawsuit-banner-2.webp)

