Reckitt Faces Lawsuit Over Enfamil Safety Claims and Emerging NEC Litigation Date: July 21, 2025
Caption: Elevator Constructors Union Local No. 1 Annuity & 401(k) Fund v. Reckitt Benckiser Group PLC, et al.
Case No.: 1:25-cv-04708
Jurisdiction: U.S. District Court, Southern District of New York
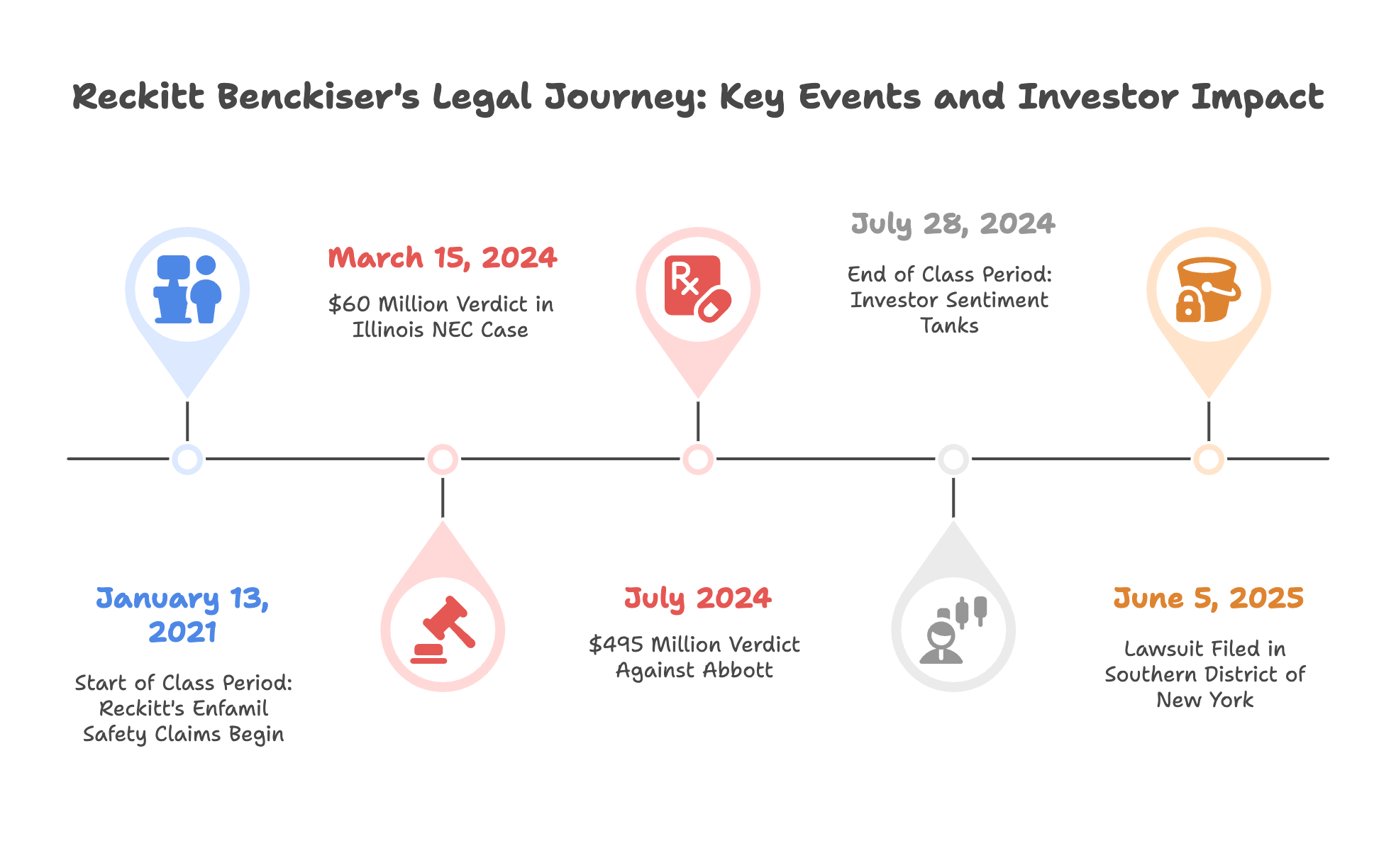
Filed: June 5, 2025
Class Period: January 13, 2021 – July 28, 2024
Introduction to Reckitt Benckiser (RBGLY) Lawsuit
Reckitt Benckiser, the U.K.-based conglomerate known for brands like Lysol, Durex, and Enfamil, is now the target of a federal securities class action. The case was filed last month in the Southern District of New York. Plaintiffs allege that Reckitt, along with several top executives, misled investors about the safety of its infant formula products and failed to disclose mounting legal risk.
The product in question is Enfamil—a flagship brand in Reckitt’s Nutrition segment and one of the most widely used infant formulas in the United States. The lawsuit claims that between 2021 and 2024, Reckitt consistently reassured investors about Enfamil’s safety and scientific backing, even as litigation over a serious condition known as necrotizing enterocolitis (NEC) was building behind the scenes.
According to the complaint, Reckitt’s statements painted a confident picture—one that left out meaningful details about Enfamil’s risks for preterm infants and the company’s growing legal exposure. That narrative began to unravel in 2024, when back-to-back jury verdicts in NEC-related cases triggered a market backlash.

Backdrop and Business Context
Reckitt is no small player. With operations in over 60 countries, its global footprint spans hygiene, over-the-counter health, and nutrition products. Its Nutrition arm, built around the $19.7 billion acquisition of Mead Johnson in 2017, includes baby formulas marketed as science-driven, clinically tested, and trusted by pediatricians.
Enfamil, the most well-known brand in the portfolio, has long held a dominant position in U.S. hospitals and retail markets. The company often described it as the “number one pediatrician-recommended” infant formula. Its advertising leaned heavily on claims of supporting immunity, digestion, and cognitive development.
But NEC—a potentially fatal intestinal condition in premature infants—has complicated that narrative. Over the past decade, clinical studies have suggested a link between cow’s milk-based formulas and NEC risk in preemies. While human milk and donor milk alternatives have gained ground, Enfamil continued to be marketed aggressively, including for vulnerable neonatal populations.
And during this same period, lawsuits alleging product liability and failure to warn were beginning to gather steam. According to the complaint, Reckitt didn’t adjust its disclosures accordingly.
Promises Made vs. Reality
The lawsuit outlines a series of public statements made by Reckitt leadership over the three-and-a-half-year class period. These statements, taken together, created an image of Enfamil as both scientifically advanced and clinically safe.
In January 2021, then-CEO Laxman Narasimhan spoke at the JPMorgan Healthcare Conference. He credited the Mead Johnson acquisition with bolstering Reckitt’s scientific credibility. He singled out Enfamil’s role in expanding the company’s R&D capabilities and digital reach. That message—of formula backed by science and scale—would be repeated for years.
In 2022, Narasimhan called product quality the “most important measure” of Reckitt’s Nutrition business. In 2023, Patrick Sly, then President of Global Nutrition, described Enfamil as the “#1 most trusted brand” among both parents and healthcare providers. CFO Jeffery Carr added that mothers were “sticking with Enfamil” and praised the brand’s momentum.
By 2024, new CEO Kristoffer Loe Licht had taken the baton. At the February Consumer Analyst Group of New York conference, he claimed Reckitt had helped resolve the U.S. infant formula shortage by delivering “40% more safe, high-quality formula.” Enfamil NeuroPro, he said, was delivering “better health and cognitive outcomes.” He credited “superior science” as the reason.
None of these statements, according to the complaint, disclosed that Reckitt was already facing NEC-related lawsuits—or that the company was aware of research linking cow’s milk-based formula to higher NEC risks in premature infants.
On April 24, 2024, Licht directly addressed the litigation. “We strongly reject any assertion that our products cause necrotizing enterocolitis,” he said. “The science does not support a causal connection.” He also insisted that Enfamil was “life-saving.” The complaint argues this response downplayed both the medical literature and the legal stakes.
Timeline of Alleged Misconduct and Disclosures
The lawsuit begins in January 2021 and runs through July 2024. In that time, Reckitt executives spoke publicly about Enfamil dozens of times—on earnings calls, at investor conferences, and in annual reports.
During this stretch:
Internal R&D efforts were repeatedly referenced as proof of product quality.
Enfamil’s market share was emphasized, especially in the non-WIC (non-government subsidized) segment.
The brand’s scientific credibility and pediatrician endorsement were promoted at every major investor touchpoint.
What was not discussed, according to the plaintiffs, was the growing wave of litigation related to NEC. In March 2024, that silence became harder to sustain.
On March 15, 2024, a jury in Illinois returned a $60 million verdict in a case alleging that Mead Johnson’s formula contributed to a premature infant’s death from NEC. Though the company denied liability, the verdict drew immediate attention. Reckitt’s stock dropped nearly 14% that day.
Then, in July 2024, a separate jury in Missouri delivered a $495 million verdict—this time against Abbott Laboratories, the maker of Similac. Still, the impact rippled. Reckitt’s shares fell another 9% the next day.
Investors, the complaint argues, had been operating under a very different impression until then.
Investor Harm and Market Reaction
For shareholders who had bought Reckitt ADSs during the class period, the losses came fast. The plaintiff fund, for instance, purchased shares at prices as high as $18.29 in 2021 and $16.27 in 2023. By the end of July 2024, the stock was trading closer to $10.
Some of that loss, of course, was driven by external factors—the verdicts, the headlines, the volatility of litigation-sensitive industries. But according to the complaint, what turned a risk into a loss was Reckitt’s failure to be candid about what it knew and when.
The verdicts didn’t create the NEC link. They revealed how little had been said about it.
Litigation and Procedural Posture
The lawsuit names Reckitt and five executives: Narasimhan, Durante, Licht, Sly, and Carr. All are accused of making false or misleading statements under Section 10(b) and Rule 10b-5. Section 20(a) “control person” claims are also included.
The core allegation is this: Reckitt knew—or had enough information to know—that Enfamil carried heightened NEC risks in preterm infants, and that litigation exposure was growing. Despite that, the company’s public statements painted a picture of clinical certainty and brand strength.
The complaint was filed June 5, 2025. Class certification and motions practice are still ahead.
Shareholder Sentiment
Before July 2024, retail investors were upbeat about Reckitt. A Stocktwits post from March 2023 cheered, “RBGLY’s Enfamil killing it—#1 brand, solid buy.” Reddit users in consumer goods threads praised Reckitt’s market share, with one writing in April 2024, “Enfamil’s pediatrician nod makes this a safe bet.” After the NEC litigation news, sentiment tanked. A July 28, 2024, Stocktwits user fumed, “They hid the NEC risk—stock’s trashed.”
On Reddit, another posted, “Reckitt knew about lawsuits and said nothing. We’re screwed.” X users echoed the anger: “$RBGLY’s silence on Enfamil risks burned us bad.” The mood shifted from trust in Reckitt’s brand to fury over perceived deception, with some investors debating whether to dump shares.
Analyst Commentary
Pre-July 2024, analysts were cautiously bullish. In early 2023, firms like Barclays set targets around $18, citing Enfamil’s U.S. dominance. Post-April 2024, after Licht’s NEC denial, analysts like Morgan Stanley held steady, projecting stability based on pediatrician trust. After the litigation fallout, the tone flipped. Barclays likely slashed targets to $10–$12, warning of “significant legal overhang.” Morgan Stanley noted brand equity risks, cutting forecasts as competitors gained ground. The shift reflected a loss of faith in Reckitt’s transparency, with analysts flagging litigation as a long-term drag on growth.
SEC Filings & Risk Factors
Throughout the class period, Reckitt filed regular reports with the SEC as required for companies trading ADSs in the U.S. These included 10-Ks, 10-Qs, and earnings supplements.
According to the complaint, these filings emphasized Enfamil’s growth, innovation, and clinical credibility. The February 2024 annual report reiterated the brand’s “science-backed” status and its #1 ranking among pediatricians.
What was missing: specific disclosures about NEC, the lawsuits, or the potential for liability.
Generic risk factors were included—things like regulatory changes, competitive threats, and supply disruptions—but none addressed the actual issue that would drive the stock down months later.
Conclusion: Investor Implications
For long-term holders, this case is more than a legal skirmish. It’s a credibility test. When a company says a product is safe, and science-based, and endorsed by doctors—you want to believe that. And when litigation arises, you expect to hear about it.
Reckitt’s lawsuit is a reminder that reputations are built on disclosure, not just performance. It also raises the stakes for companies selling products in sensitive categories like infant health. If risks emerge, silence may not be strategic—it may be actionable.
The lawsuit is still playing out. But for investors, the lesson is already clear: marketing is one thing. Risk disclosure is another. And when they don’t line up, it’s the shareholders who end up paying the difference.



![Reckitt Benckiser Group PLC (RBGLY) Securities Class Action Lawsuit Update [July 7, 2025]](https://dropinblog.net/cdn-cgi/image/fit=scale-down,format=auto,width=700/34260815/files/featured/reckitt-benckiser-group-lawsuit-blog-banner.webp)