Hims Faces Lawsuit Over Knockoff Wegovy and Novo Nordisk Split July 21, 2025
Caption: Sookdeo v. Hims & Hers Health, Inc., et al.
Case No.: 3:25-cv-05315
Jurisdiction: U.S. District Court, Northern District of California
Filed on: June 25, 2025
Class Period: April 29, 2025, to June 23, 2025

Introduction to Hims & Hers Health (HIMS) Securities Lawsuit
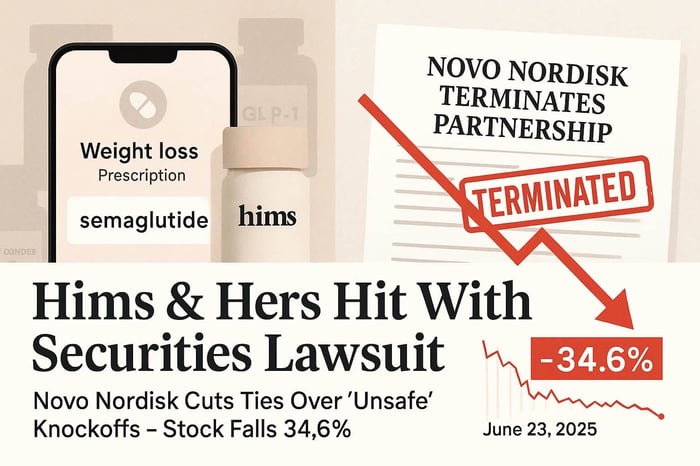
Hims & Hers Health, Inc. (NYSE: HIMS) is in the crosshairs. A class action lawsuit landed in California's Northern District on June 25, 2025. It accuses Hims and execs (CEO Andrew Dudum and CFO Yemi Okupe) of misleading people who bought stock from May 5 to June 23, 2025. Plaintiff Michael Sookdeo says Hims pushed knockoff Wegovy as safe and personalized, but Novo Nordisk yanked the partnership, calling it deceptive and risky. The stock crashed 34.6% on June 23, 2025, from $64.22 to $41.98. This unpacks the claims, Hims' setup, the investor hit, and lessons for health stock watchers.
Backdrop and Business Context
Hims sells telehealth meds for hair loss, ED, and weight loss. Its model skips docs for direct access to generics and compounds. . Hims' revenue hinges on subscribers. Subscribers were up 41% in Q1 2025, to 1.7 million. In 2025, Hims jumped on the GLP-1 craze, partnering with Novo Nordisk for compounded semaglutide, a Wegovy knockoff, amid shortages. They touted it as personalized, safe, and affordable, blending it with nutrition kits and care plans. But compounded drugs are tricky and FDA says they're not approved. So, safety's a gamble if not done right.
Promises Made vs. Reality
Hims' brass painted compounded GLP-1s as a game-changer. On April 29, 2025, a press release hailed the Novo Nordisk tie-up for "affordable access" to semaglutide, calling it "high-quality" and "personalized." Dudum said it tackled shortages while "prioritizing safety." The Q1 earnings call echoed this, with Dudum bragging about "innovative partnerships" for consumer choice. The shareholder letter pushed oral kits and compounded GLP-1s as safe starters.
The lawsuit says that was bunk. Hims hawked "illegitimate knockoffs" from shady Chinese suppliers, risking patient safety. Novo Nordisk called it deceptive mass sales under a personalization guise. Hims knew the risks but hid them, inflating stock until the partnership blew up.
Timeline of Alleged Misconduct and Disclosures
The mess ran from May 5 to June 23, 2025 (the “Class Period”).
On May 5, Hims announced the Novo Nordisk deal for compounded semaglutide, sending shares up. The Q1 call and letter hyped it as safe and personalized, keeping investors hooked. Stock climbed to $64.22 by June 20.
On June 23, Novo Nordisk dumped Hims, blasting "deceptive marketing" and "unsafe" knockoffs from China. Hims' stock tanked 34.6% to $41.98 on heavy volume.
Investor Harm and Market Reaction
The split was a bloodbath. Sookdeo, buying at inflated prices, got hammered. Analysts piled on—one slashed targets, warning of lost trust and revenue hits. The market shrugged off Hims' safety spin, with shares cratering as investors fled. Folks who bought the personalization hype paid big when Novo Nordisk called foul.
Litigation and Procedural Posture
Filed in Northern California, the suit names Hims, Dudum, and Okupe for violating Section 10(b) and Rule 10b-5, plus Section 20(a) for control. It claims they knew compounded semaglutide was risky but lied about safety. Dropped June 25, 2025, it seeks class status and jury trial. Early stages, no big moves yet, but Hims' 220 million shares point to a wide net of burned investors.
Shareholder Sentiment
Before the June 23, 2025, bombshell, Hims & Hers Health had retail investors buzzing like bees around honey. On platforms like Stocktwits, Reddit, and X, the vibe was electric. A May 10, 2025, Stocktwits post gushed, “$HIMS is killing it with GLP-1 knockoffs—Wegovy’s got nothing on this!” Over on Reddit’s r/WallStreetBets, users were hyped, with one April 2025 thread proclaiming, “Hims’ personalized meds are a game-changer. Affordable semaglutide? I’m loading up!” X posts echoed the love, with a user tweeting in early June, “$HIMS partnering with Novo Nordisk is huge—weight loss meds are gonna rocket this stock.” Investors saw Hims as a telehealth darling, cashing in on the GLP-1 craze with a slick, direct-to-consumer model that screamed growth.
Then Novo Nordisk pulled the plug, and the mood flipped. After the June 23 press release called Hims’ compounded drugs “deceptive” and “unsafe,” Stocktwits turned fiercely negative. One user raged, “They sold us on safe knockoffs, and now the stock’s trashed. Dudum’s got blood on his hands!” Reddit threads were brutal, with a June 24 post venting, “Hims knew those Chinese suppliers were sketchy—lied to our faces and screwed our wallets.” Another user added, “I bought at $60, now it’s $41. Trust’s gone, and I’m out.” On X, the fury was raw: “$HIMS hyped personalization, but it was a scam. Partnership dead, stock’s a corpse.” Some investors debated holding on, hoping for a rebound, but most were livid, feeling burned by Hims’ safety spin and ready to dump shares at the first chance. The shift from hype to betrayal was stark, with the 34.6% stock drop fueling cries of deception across the boards.
Analyst Commentary
Before the June 23 fallout, analysts were all-in on Hims. In May 2025, Bank of America set a $70 price target, crowing about the Novo Nordisk deal as a “growth rocket” for Hims’ weight loss segment. J.P. Morgan, in a June note, pegged $65, praising Hims’ “innovative” GLP-1 push and 41% subscriber growth. Piper Sandler was even rosier, slapping a $75 target in late May, saying, “Hims’ personalized compounds give it an edge in telehealth’s hottest market.” The consensus saw Hims riding the weight loss wave, with the Novo tie-up as a golden ticket to scale.
Post-June 23, the tone crashed harder than the stock. Bank of America slashed their target to $30, calling the Novo split a “trust killer” and warning of “significant revenue headwinds” without Wegovy access. J.P. Morgan cut to $35, flagging “deceptive marketing” as a blow to Hims’ brand and predicting regulatory scrutiny. Piper Sandler dropped to $32, noting, “The loss of Novo’s backing and questions about Chinese suppliers crater Hims’ credibility.” Analysts also pointed to broader fallout: the FDA’s resolved semaglutide shortage meant legit Wegovy was back, squeezing Hims’ knockoff niche. One boutique firm warned, “Hims’ pivot to other meds won’t offset the damage—investors will stay wary.” The mood swung from bullish to grim, with analysts questioning Hims’ ability to rebuild trust and navigate a tougher regulatory landscape.
SEC Filings & Risk Factors
Hims’ Q1 2025 filings hyped GLP-1 compounds as safe, personalized winners, spotlighting the Novo Nordisk deal as a growth driver. The May 5, 2025, 10-Q noted the FDA’s semaglutide shortage resolution but buried risks of compounded drugs. No whisper of Chinese suppliers or safety concerns surfaced, despite execs knowing the shaky ground.
The lawsuit claims this dodge misled investors, inflating stock prices. The June 23, 2025, 8-K finally spilled the beans on Novo’s split, citing deceptive practices, but it was too late—shares had already tanked hard.
Conclusion: Investor Implications
Hims' mess warns health stock chasers. Pushing knockoffs as safe can blow up fast. Spot red flags: vague safety claims, partnerships with big pharma that sour quick. For telehealth plays, transparency on drug sources is key. Investors wait for the suit to shake out and Hims to scramble. Dig past hype—check supply chains and regs, because in meds, a bad batch can kill your portfolio.