Introduction to Replimune Group, Inc. (REPL) Securities Lawsuit
Group, Inc. (NASDAQ: REPL) has been hit with a securities class action suit. The plaintiff says REPL’s bluster about its RP1 program and Biologics License Application were false or misleading. The case represents people who got REPL stock between November 22, 2024 through July 21, 2025, inclusive (“the Class Period”). Specifically, plaintiffs say Replimune failed to disclose key issues with its IGNYTE trial design. The FDA later rejected the BLA, and the company’s stock fell sharply. Plaintiffs claim Replimune's false and misleading statements misled investors and propped up REPL's stock price. But, once the truth was revealed, the stock crashed and investors suffered substantial losses.
Replimune Group, Inc. (REPL) Securities Case Details
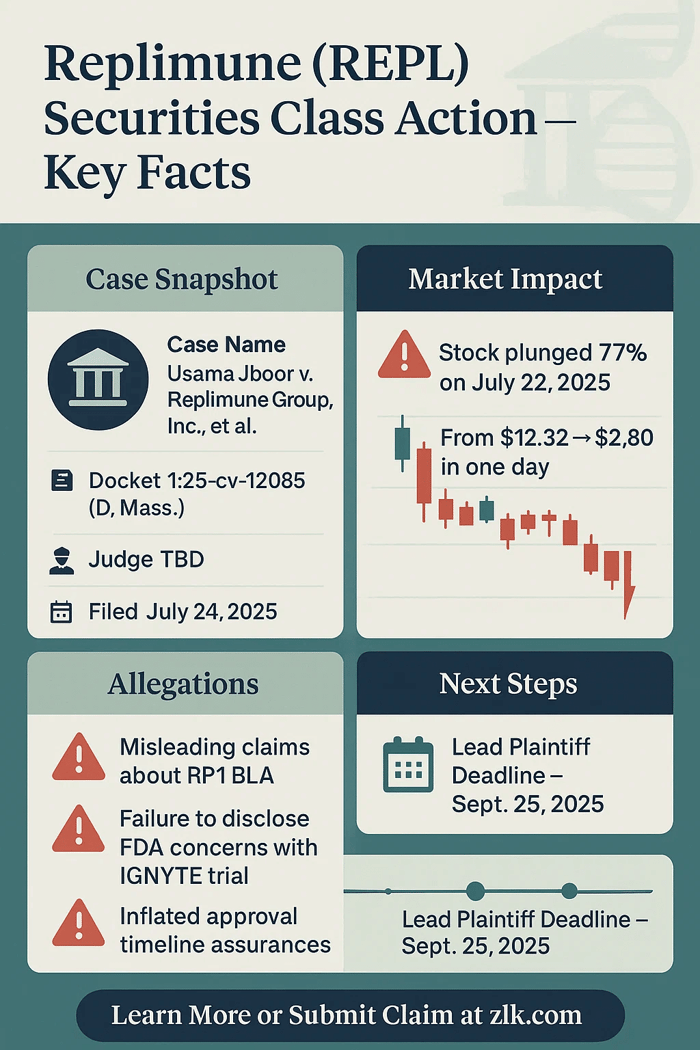
Case Name: Jboor v. Replimune Group, Inc., et al.
Docket Number: 1:25-cv-12085
Court: U.S. District Court for the District of Massachusetts
Filed: July 24, 2025
Replimune Group, Inc. (REPL) Company Profile
Replimune (NASDAQ: REPL) is a biotech company. It specializes in developing oncolytic immunotherapies (that is, genetically modified viruses that infect and destroy cancer cells). Its lead product, is RP1, which it describes as a crucial new treatment for some forms of skin cancer. The treatment is designed to kill tumors directly while activating an immune response. The Ignyte Study assessed the drug's effectiveness and safety.
Class Period
November 22, 2024 to July 21, 2025, inclusive.
Investors who got REPL stock during this time and experienced losses may be included in the Replimune securities fraud class action lawsuit.
Allegations in the Replimune Group, Inc. (REPL) Class Action Securities Lawsuit
The complaint names Replimune, its CEO Sushil Patel, and its CFO Emily Hill. Plaintiffs say company made mislead people about the progress and quality of its clinical data.
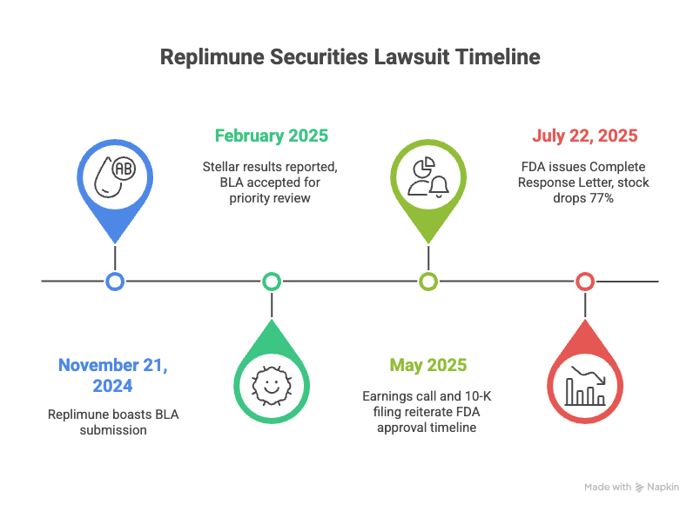
In its November 21, 2024 press release, Replimune boasted that it had sent in a BLA for RP1 to the Food and Drug Administration. In the press release, the company’s execs underscored the drug’s Breakthrough Therapy designation and insisted the trial data supported the submission.
In February 2025, the company reported stellar results: a 33.6% overall response rate and durable patient impact. The company even said the BLA had been accepted for an accelerated approval pathway.
Replimune reiterated its glowing assessment of the FDA approval timeline and data in its May 2025 earnings call and 10-K filing. The company’s top brass said all interactions with the FDA were complete, there were no known barriers, and the company was on track for the July 22, 2025 PDUFA date. But, according to plaintiffs, those statements neglected to reveal something important: the concerns raised by the FDA that might have put a wrench in the works.
The core allegation is that the company did not tell investors the RP1 trial (called “IGNYTE”) was not adequate or sufficiently well-designed, and that the FDA had serious, material issues about the trial population and design.
The Truth Emerges
On July 22, 2025, Replimune revealed the FDA had issued a Complete Response Letter. The agency said it could not approve the BLA in its current form. The FDA found the IGNYTE trial inadequate, was poorly designed, and that the data could not be interpreted with confidence. It also raised issues about the confirmatory trial and the role of each component in the treatment. There were no safety concerns, but the agency concluded the company had not proven that the treatment worked.
Market Reaction
The market reacted swiftly: shares of Replimune fell from $12.32 to $2.80 on July 22, 2025. That represented a drop of about 77% on unusually high volume.
Next Steps
Submissions for lead plaintiff are due September 25, 2025.
The Court will issue its order for lead plaintiff and counsel in the weeks after lead plaintiff submissions are due.
The Court will then consider motion for class certification.
The Court will later consider a Motion to Dismiss.
To learn if you are eligible for recovery under the Replimune group securities class action lawsuit, visit the case submission page here.
Disclaimer: This shareholder alert is for informational purposes only and does not constitute legal advice. Consult a qualified attorney for personalized guidance. No specific outcomes are guaranteed. Speak with a licensed attorney for guidance specific to your situation.
![Replimune Group, Inc. (REPL) Securities Class Action Lawsuit [September 8, 2025]](https://dropinblog.net/cdn-cgi/image/fit=scale-down,format=auto,width=700/34260815/files/featured/replimune-securities-lawsuit-blog-banner-v2.webp)


![RxSight, Inc. (RXST) Securities Class Action Lawsuit Update [September 03, 2025]](https://dropinblog.net/cdn-cgi/image/fit=scale-down,format=auto,width=700/34260815/files/featured/rxsight-securities-lawsuit-blog-banner.webp)
![Alto Neuroscience, Inc. (ANRO) Securities Class Action Lawsuit [September 02, 2025]](https://dropinblog.net/cdn-cgi/image/fit=scale-down,format=auto,width=700/34260815/files/featured/altoneuroscience-securities-lawsuit-blog-banner.webp)