Introduction to Rocket Pharmaceuticals, Inc. (RCKT) Lawsuit
A securities fraud class action lawsuit has been filed against Rocket Pharmaceuticals, Inc. (NASDAQ: RCKT) on behalf of investors who purchased the Company’s common stock between February 27, 2025, and May 26, 2025 (the “Class Period”). The lawsuit alleges that Rocket and certain executives violated federal securities laws by making statements about the company’s lead drug trial that left out critical risks—specifically, changes to the trial protocol that were never disclosed until after a patient died and the FDA intervened.
Rocket Pharmaceuticals, Inc. (RCKT) Securities Lawsuit Case Details
Caption: Ho v. Rocket Pharmaceuticals, Inc., et al.
Case No.: 3:25-cv-10049
Jurisdiction: U.S. District Court, District of New Jersey
Filed on: June 11, 2025 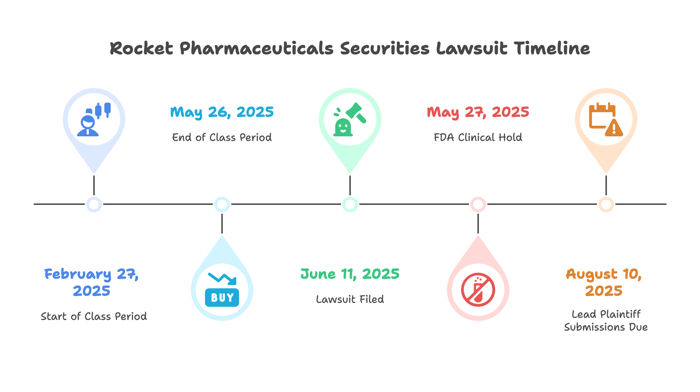
Rocket Pharmaceuticals, Inc. (RCKT) Company Profile
Rocket is a biotech company based in New Jersey. It focuses on gene therapies for rare inherited diseases. One of its most advanced candidates is RP-A501, a treatment for Danon disease. Danon disease is a serious pediatric heart condition. The company launched a Phase 2 trial studying the safety and efficacy of RP-A501.

Class Period:
February 27, 2025 – May 26, 2025
Investors who purchased Rocket securities during the class period might be eligible to join the Rocket Pharmaceuticals securities class action lawsuit.
Allegations in the Rocket Pharmaceuticals, Inc. (RCKT) Lawsuit
The complaint says Rocket misled investors by omitting a significant change to the RP-A501 clinical trial. Specifically, the company reportedly added a new immunosuppressive drug to its protocol months before publicly disclosing that change. The added drug, a C3 inhibitor, was intended to address earlier safety issues.
But public statements during this time continued to highlight safety and progress. In a February press release and again in a May earnings call, Rocket described the trial as ongoing, referenced strong earlier data, and gave no hint of internal concerns.
Plaintiffs say those statements were incomplete and misleading. Two patients received the revised protocol and both developed capillary leak syndrome. One of them later died. The complaint claims Rocket waited to reveal these facts until the FDA had already issued a clinical hold.
The filed complaint alleges Rocket’s failure to disclose these risks violated federal securities laws, including Sections 10(b) and 20(a) of the Securities Exchange Act. Plaintiffs’ legal claims state that Rocket’s statements about RP-A501’s safety and trial timeline were materially false and misleading, lacking a reasonable basis, and adversely affected investors.
The Truth Emerges
On May 27, 2025, Rocket issued a press release stating FDA put a halt to the RP-A501 trial. The company confirmed that a patient died after receiving the newly added immune suppression agent. During a same-day investor call, CEO Gaurav Shah explained that the protocol amendment had been made “several months” earlier but withheld from investors. He acknowledged that two patients were affected by the change and said the company was investigating the possible connection between the drug and the adverse events.
Market Reaction
Rocket’s stock dropped 37% on the news, falling from $6.27 to $3.33 per share. The decline reflected not just the loss of confidence in the program, but the sudden shock of information that had been withheld. J.P. Morgan issued a note calling the clinical hold “a major setback” and questioned whether the trial could recover. Cantor Fitzgerald lowered its price target from $30 to $10. Both pointed to concerns about safety and transparency.
Next Steps
Submissions for lead plaintiff are due August 10, 2025.
The Court will issue its order for lead plaintiff and counsel in the weeks after submissions are due.
The Court will then consider motion for class certification.
The Court will later consider a Motion to Dismiss.
To learn if you are eligible for recovery under the Rocket Pharmaceuticals securities class action lawsuit, visit the case submission page here.
Disclaimer: This shareholder alert is for informational purposes only and does not constitute legal advice. Consult a qualified attorney for personalized guidance. No specific outcomes are guaranteed.
![Rocket Pharmaceuticals, Inc. (RCKT) Securities Class Action Lawsuit Update [July 16, 2025]](https://dropinblog.net/cdn-cgi/image/fit=scale-down,format=auto,width=700/34260815/files/featured/rocket-pharma-rckt-securities-lawsuit-blog-banner.webp)



![iRobot Corporation (IRBT) Securities Class Action Lawsuit Update [August 15, 2025]](https://dropinblog.net/cdn-cgi/image/fit=scale-down,format=auto,width=700/34260815/files/featured/irobot-securities-lawsuit-blog-banner.webp)