Rocket Faces Legal Heat Over Hidden Risks in Gene Therapy Trial
Caption: Ho v. Rocket Pharmaceuticals, Inc., et al.
Case No.: 3:25-cv-10049
Jurisdiction: U.S. District Court, District of New Jersey
Filed: June 11, 2025
Class Period: February 27, 2025 – May 26, 2025
Introduction to Rocket Pharmaceuticals, Inc. (RCKT) Securities Class Action Lawsuit
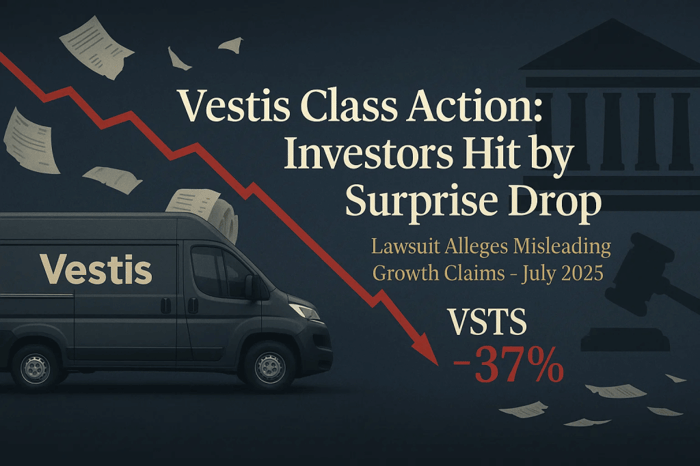
Rocket Pharmaceuticals, Inc. (NASDAQ: RCKT) is under fire. On June 11, 2025, a federal securities class action lawsuit was filed in New Jersey. It accuses the company and CEO Gaurav Shah of misleading investors about the safety of a clinical trial protocol. The case centers on statements made between late February and late May of this year, just before a sudden disclosure of a patient death and an FDA-imposed clinical hold sent the company’s stock into a 37% tailspin.
The lead plaintiff, Rondolph Ho, claims that Rocket concealed a key risk: a quiet change to a clinical trial protocol that introduced a new immunosuppressive drug to the study. This change led to serious adverse events and ultimately proved fatal for a participant in the trial. The lawsuit argues that Rocket Pharmaceuticals knew about this change and its potential consequences months before the news broke, which constitutes alleged securities fraud as the company failed to timely inform investors.
This report walks through the business background, the timeline of public and private disclosures, the fallout for shareholders, and what this case may signal for investors in biotech companies navigating similar terrain.

Backdrop and Business Context
Rocket Pharmaceuticals is a clinical-stage biotech company. Its focuses on gene therapies for rare, life-threatening diseases affecting children and young adults. The company’s lead program, RP-A501, is designed to treat Danon disease. Danon disease is rare genetic cardiac disorder. Most patients diagnosed with Danon are young males. Without intervention, many progress rapidly to heart failure.
The science behind Rocket’s approach is complex, but the business pitch is straightforward: introduce a novel immunomodulatory gene therapy dose that offers years—possibly decades—of clinical benefit. RP-A501 uses adeno-associated virus (AAV) vectors to transport healthy genes into cells. That delivery system isn’t new, but it comes with risks. Among them: triggering an immune system overreaction called complement activation. If not managed properly, it can cause severe adverse events.
Rocket completed an early-stage trial in 2022 and moved to a larger Phase 2 trial in 2023. The company signaled that RP-A501 was heading toward FDA approval by 2026.
Promises Made vs. Reality
Rocket made overwhelmingly positive statements in early 2025 and leaned hard on its supposed progress. On February 27, the company issued its annual results. Shah praised RP-A501’s safety and long-term efficacy based on Phase 1 data published in the New England Journal of Medicine. He told investors that things were on track. A program update was coming mid-year. Data from the pivotal trial was expected in 2026.
Then came the May 8 earnings call. Shah again underscored the momentum. He highlighted another study—this one in the Journal of the American Heart Association—that emphasized the unmet need in Danon disease. The company, he said, was well-positioned to deliver.
But the lawsuit claims those assurances left out a crucial detail. Months earlier, Rocket had modified the trial protocol. They added a new drug: a complement C3 inhibitor, designed to suppress immune flare-ups during treatment. This change wasn’t disclosed to shareholders. The company didn’t mention it in filings, on calls, or in investor presentations.
Two participants received the new drug regimen. Both developed capillary leak syndrome, a serious immune reaction. One of them died from complications related to a follow-on infection. On May 23, the FDA placed the trial on hold. Rocket disclosed the news on May 27. Until then, investors had no reason to suspect anything was off course.
Timeline of Alleged Misconduct and Disclosures
The Rocket Pharmaceuticals securities class action lawsuit focuses on a three-month window between late February and late May. During this time, Rocket repeatedly issued positive statements about its lead therapy and the pace of its clinical work.
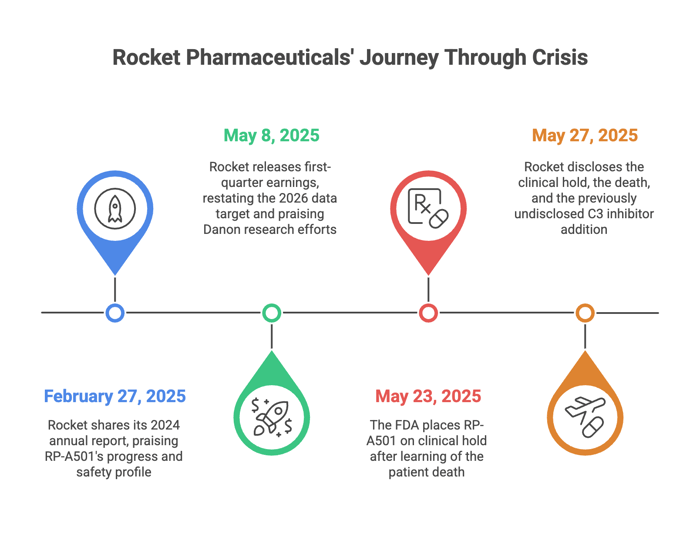
February 27, 2025: Rocket shares its 2024 annual report. Shah praises RP-A501’s progress and safety profile. The stock holds steady.
March–April 2025: Investors post hopeful takes on the company’s outlook, citing both published data and the upcoming program update.
May 8, 2025: Rocket releases first-quarter earnings. Shah restates the 2026 data target and praises Danon research efforts. No mention of safety concerns or changes to the treatment protocol.
May 23, 2025: The FDA places RP-A501 on clinical hold after learning of the patient death.
May 27, 2025: Rocket issues an 8-K disclosing the hold, the death, and—only then—the previously undisclosed C3 inhibitor addition. Shah states the change was made “several months ago.”
By the time the disclosure landed, Rocket’s stock had fallen from $6.27 to $2.33.
Investor Harm and Market Reaction
The market didn’t take the news quietly. Rocket Pharmaceutical securities plunged 37% on May 27 alone, wiping out over a third of Rocket’s market value. For investors, the losses were swift and deep.
Analysts moved quickly. J.P. Morgan called the disclosure a “major blow,” slashing their target price and raising concerns about Rocket’s transparency. Cantor Fitzgerald echoed the concern, noting that the clinical hold could delay FDA approval by years. Leerink Partners and TD Cowen both downgraded the stock.
Litigation and Procedural Posture
The complaint, filed in the District of New Jersey, alleges violations of federal securities laws, namely Section 10(b) of the Exchange Act and SEC Rule 10b-5. It also includes a claim under Section 20(a) of the Exchange Act, naming CEO Gaurav Shah as a controlling person. The central claim is that Rocket misled investors throughout the class period by failing to disclose the protocol change, despite knowing it carried clinical risk.
Plaintiffs argue that the company’s leadership had a duty to inform the market—especially once patients suffered adverse effects. The fact that the C3 inhibitor was introduced quietly and not mentioned until after the death and FDA action forms the backbone of the case. That is, plaintiffs say defendants withheld material adverse facts from shareholders despite knowing of those facts and thus artificially inflated prices of the stock.
No major court rulings have occurred yet. The case seeks class certification and a jury trial. Early discovery may focus on internal documents surrounding the timing and rationale of the protocol change.
Shareholder Sentiment
Before the May 27 disclosure, investor discussion around Rocket was cautiously optimistic. Retail shareholders, particularly on biotech-focused forums, pointed to early success and credible scientific backing. Some had followed the company since its IPO. Others had bought in more recently, drawn by the potential upside of a successful Danon therapy.
A March post on Stocktwits read: “This is a make-or-break stock, but if RP-A501 delivers, it changes lives. Holding long.” On Reddit, a user commented in April: “Data looks solid. Danon’s a tough disease—this is real innovation.”
But the mood flipped fast. After the May disclosure, threads filled with frustration. “They knew and said nothing,” one user posted. Another wrote, “The death is tragic, but the betrayal is what stings. Investors weren’t warned.” On X, one post summed up the tone: “C3 change was a major shift. Silence wasn’t just risky—it was reckless.” Some holders said they planned to exit. Others stayed in, hoping for recovery—but with lowered expectations and higher skepticism.
Analyst Commentary
Prior to May 27, analysts were still backing the company—though with some reservations. J.P. Morgan had a $9 target in February, citing strong Phase 1 data. Cantor Fitzgerald listed a target near $8.50, flagging the inherent risks of AAV therapies but praising Rocket’s focus. Leerink’s March report cited the company’s niche focus and projected an inflection point by mid-2025.
After the FDA hold, coverage changed quickly. J.P. Morgan dropped its target to $4 and noted that Rocket’s communication strategy raised concerns. Cantor cut to $4.50. TD Cowen, which had maintained a more neutral stance, downgraded its rating and warned about long-term brand erosion.
A common thread in all the reports: the concern wasn’t just the safety event, but the way information had been managed. Analysts warned that investor confidence—especially in biotech—was slow to rebuild once lost.
SEC Filings & Risk Factors
Between February and May 2025, Rocket filed multiple reports with the SEC. The company’s 10-K, dated February 27, included broad risk language around immune responses and trial challenges. But it didn’t reference the addition of the C3 inhibitor or any specific changes to the treatment protocol.
The company’s quarterly 10-Q followed a similar pattern. The filings discussed Danon disease, the RP-A501 trial, and general industry risks. But the key change—the one that introduced a new immunosuppressive drug—was not flagged.
It wasn’t until May 27 that Rocket filed an 8-K disclosing the death, the clinical hold, and the protocol change. The lawsuit points to this as the crux of the issue: that for months, investors were told the trial was progressing normally, when internally, that was no longer true.
Conclusion: Investor Implications
The Rocket Pharmaceuticals case isn’t just about a failed disclosure. It’s about how biotech companies walk the line between optimism and transparency. Clinical trials—especially in gene therapy—carry known risks. What’s less forgivable is silence. For investors, the lesson is straightforward. Look beyond scientific promise. Pay close attention to how companies handle setbacks, adjust trial protocols, and manage expectations. Executive tone can shape the market, but absence of detail often says more.
The lawsuit is still in early stages. But its outcome may influence how biotech firms treat risk reporting in investor materials. For Rocket, the road to regaining trust will likely be longer than the path to resolving its clinical hold.


![Rocket Pharmaceuticals, Inc. (RCKT) Securities Class Action Lawsuit Update [July 16, 2025]](https://dropinblog.net/cdn-cgi/image/fit=scale-down,format=auto,width=700/34260815/files/featured/rocket-pharma-rckt-securities-lawsuit-blog-banner.webp)