Introduction to Viatris, Inc. (VTRS) Lawsuit
This is a shareholder alert about the Viatris, Inc. class action lawsuit filed on or about April 4, 2025. The lawsuit seeks to recover losses of shareholders adversely affected by alleged securities fraud. The Viatris lawsuit seeks to represent purchasers of Viatris, Inc. securities during the class period, which is between August 8, 2024 to February 26, 2025. Shareholders who suffered a loss during the class period are eligible to participate in the lawsuit without needing to be appointed as lead plaintiff. The deadline to file for lead plaintiff in the Viatris securities fraud class action lawsuit is June 3, 2025.
Viatris is a pharmaceutical company with facilities worldwide.
In December 2024, Viatris announced it received an FDA warning letter because its Indore, India facility failed inspection. Despite this failure, Defendants routinely referred to remediation efforts it was making and that it did not anticipate the Indore Facility Warning Letter impacting Viatris’ 2024 guidance ranges. Later, the Company further downplayed the Warning Letter because the Indore Facility was only 1 of 26 company facilities.
However, in February 2025, the Company released its 2025 fiscal guidance and updated its 10-K SEC filing. During the associated earnings call, Company executives revealed substantial headwinds due to the problems at the Indore facility, with negative revenue totality $500 million.
Investors reactive negatively and Viatris’ common stock price plunged about 15% in one day.
Viatris, Inc. (VTRS) Lawsuit Case Details
Quinn v. Viatris, Inc., et al Case No. 2:25-cv-00466
U.S. District Court, Western District of Pennsylvania
Filed on April 4, 2025
Viatris, Inc. (VTRS) Company Profile
Viatris is a pharmaceutical and global healthcare company formed by a merger of Myland and Upjohn in 2020. It offers prescription brand-name drugs and generic drugs. Some of its major projects include Lipitor, Celebrex, Viagra, EpiPen, and Zoloft, amongst others. Viatris maintains facilities and does business worldwide
Viatris, Inc. (VTRS) Securities Lawsuit Class Period
August 8, 2025 to February 26, 2025, inclusive.
Investors who acquired Viatris securities during the class period may be eligible for compensation from the class action lawsuit.
Viatris, Inc. (VTRS) Securities Allegations
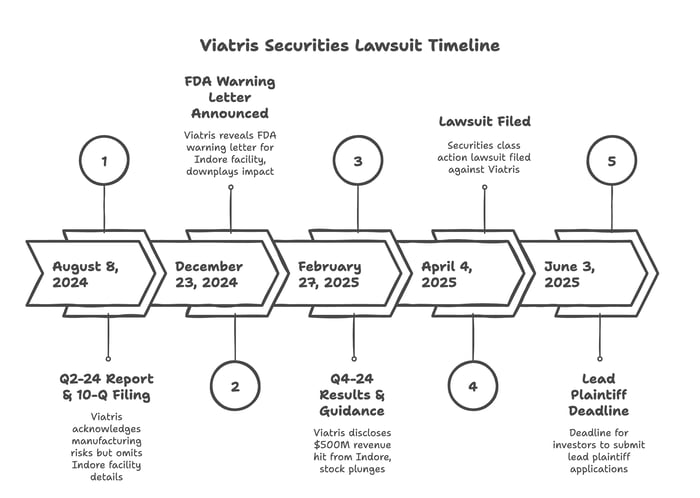
On August 8, 2024 Viatris issued its Q2-24 financial report and filed its SEC Form 10-Q. The 10-Q described major company risks including “changes in or difficulties with the Company’s manufacturing facilities including with respect to inspections, remediation and restructuring activities . . .” The Company made an identical statement on its Q3-24 filing. Despite this acknowledged risk factor, Defendants did not mention any inspection of its Indore, India facility in its Q2-24 filings or associated earnings call.
On December 23, 2024, Viatris issued a press release stating the FDA issued a warning letter after a failed inspection of the Company’s Indore, India facility. The letter stated, in part, “the Agency has issued a warning letter, and an Import Alert related to this facility.” Because of the warning, the Company admitted “11 actively distributed products” would no longer be accepted into the U.S. until the FDA lifted the Warning Letter. Four products on the list were on “shortage concerns” so Viatris noted there “could be potential for additional exceptions based on further discussions with the Agency.”
The letter noted Viatris first learned of the inspection results at an undisclosed time earlier in the year. Since then, the Company made ongoing remediation efforts. Despite the news, the Company, Viatris emphasized “we do not anticipate these actions impacting our current 2024 guidance ranges.”
The Company continued to downplay the impact of the Warning Letter at a January 2025 J.P. Morgan Health Conference. There, Company executives highlighted the Indore facility is “1 of 26 facilities” and therefore the expected financial impact of the FDA letter was likely minimal.
The Complaint filed against Viatris accuses the Company and some of its executives of securities fraud, in violation of federal securities laws, such as the Securities Exchange Act. Plaintiffs allege Defendants provided investors false, misleading, or materially inaccurate information about the Company's Indore, India facility. The Complaint alleges Defendants' statements misrepresented and downplayed the financial impacted of the FDA Letter on the Company’s finances.
The Truth Emerges
On February 27, 2025 Defendants released the Fourth Quarter 2024 financial results and 2025 fiscal guidance. Defendants also updated their 10-K filing and provided disclosures regarding the FDA inspection of the Indore facility.
During the associated earnings call, Company executives revealed the Indore inspection occurred in June 2024 and no other exceptions would be granted, despite Viatris’ previous suggestions otherwise. They also revealed substantial headwinds due to these efforts, with total negative revenue of approximately $500 million.
Market Reaction
Investors reacted negatively to this news. Viatris' stock price fell 15.21% below its closing market price prior to these public statements.
Next Steps
Submissions for possible lead plaintiff appointment are due June 3, 2025.
The Court will issue its order for lead plaintiff and counsel in the weeks after submissions are due.
The Court will then consider motion for class certification.
The Court will later consider a Motion to Dismiss.
To learn whether you may be eligible for a recovery under this class action, go to the case submission page found here.
![Viatris, Inc. (VTRS) Securities Class Action Lawsuit Update [May 2, 2025]](https://dropinblog.net/cdn-cgi/image/fit=scale-down,format=auto,width=700/34260815/files/featured/8d5a6d43-1300-488e-bb0e-7b5390d5ac2d.webp)

![Everus Construction Group, Inc. (ECG) Securities Class Action Lawsuit Update [May 6, 2025]](https://dropinblog.net/cdn-cgi/image/fit=scale-down,format=auto,width=700/34260815/files/featured/79861259-43b2-4a28-94dd-b14fd46d36f9.jpeg)

![Sana Biotechnology, Inc. (SANA) Securities Class Action Lawsuit Update [April 21, 2025]](https://dropinblog.net/cdn-cgi/image/fit=scale-down,format=auto,width=700/34260815/files/featured/sana-biotechnology-lawsuit.webp)