Introduction
Sarepta Therapeutics, Inc. (NASDAQ: SRPT) is facing a shareholder lawsuit after safety concerns surfaced surrounding its gene therapy ELEVIDYS. Filed in the Southern District of New York, the complaint targets the company’s public statements between June 22, 2023, and June 24, 2025—a period during which Sarepta told investors one version of events, while a different story appears to have played out behind closed doors.
Case Details
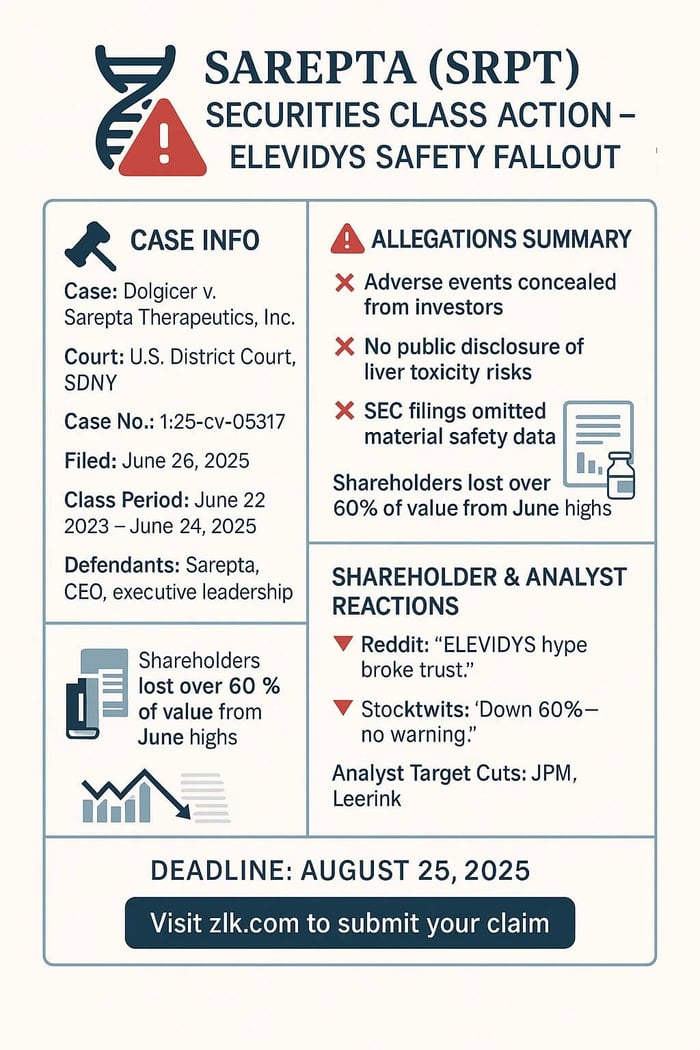
Caption: Dolgicer v. Sarepta Therapeutics, Inc., et al.
Case No.: 1:25-cv-05317
Court: U.S. District Court, Southern District of New York
Filed: June 26, 2025
Backdrop and Business Context
Sarepta is a biotech company headquartered in Massachusetts that focuses on genetic medicines for rare diseases. Its primary product, ELEVIDYS, is a gene therapy designed to treat children with Duchenne muscular dystrophy (DMD), a fatal muscle-wasting disorder. For patients and investors alike, it represented hope.
During the class period, Sarepta leaned heavily on ELEVIDYS. It was the centerpiece of investor presentations, earnings calls, and press releases. By late 2024, the company had reported more than $1 billion in cumulative revenue from the drug.
Promises Made vs. Reality
Sarepta claimed ELEVIDYS was safe, effective, and making strong inroads in both ambulatory and non-ambulatory DMD patients. The FDA had accepted its updated Biologics License Application, and the company said access would expand to children aged four and older.
But according to the lawsuit, that optimism masked a growing safety issue. The company allegedly knew about serious adverse events linked to ELEVIDYS—some involving liver damage—but did not disclose them in real time. Two patients died. Regulators intervened. Trials were paused. Shipments were suspended. Yet none of that was shared with the public until much later.
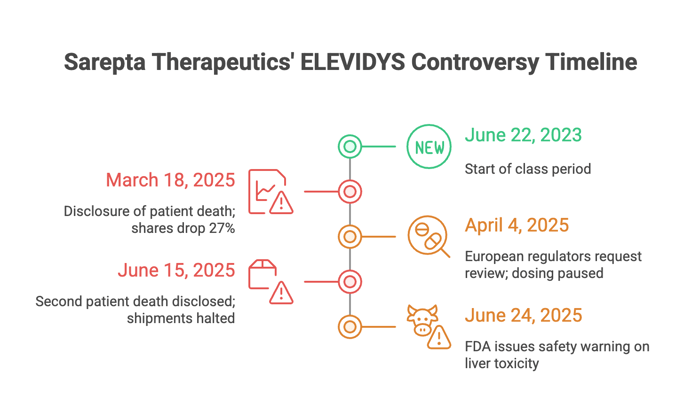
Timeline of Alleged Misconduct and Disclosures
- March 18, 2025: Sarepta disclosed a patient death connected to ELEVIDYS. Shares plunged 27%.
- April 4, 2025: European regulators requested a full review. Sarepta paused dosing. Stock fell again.
- June 15, 2025: A second patient death was made public. Shipments were halted for certain patients.
- June 24, 2025: The FDA issued a safety warning. Liver toxicity was now a known concern.
The lawsuit argues these events weren’t isolated accidents—they were known risks that Sarepta failed to flag.
Investor Harm and Market Reaction
The consequences for shareholders were severe:
- March 18: –27.44% to $73.54
- April 4: –7.13% to $54.43
- June 15: –42.12% to $20.91
- June 25: –8.01% to $17.46
Investors say these cascading drops didn’t come from general biotech volatility—but from disclosures that should’ve come months earlier.
Litigation and Procedural Posture
Filed on June 26, 2025, the complaint alleges that Sarepta violated Sections 10(b) and 20(a) of the Securities Exchange Act. Specifically, that Sarepta knowingly or recklessly made misleading statements, and that its executives had a duty to update the market when safety concerns arose.
Next, the court will appoint a lead plaintiff and legal counsel. After that, motions to dismiss and class certification will be on the table. If the case survives, discovery could reveal internal documents and communications that shed light on what Sarepta’s leadership knew—and when.
Shareholder Sentiment
In the early months of the class period, sentiment was high. Reddit and Stocktwits were filled with optimism. “SRPT’s a moonshot,” one user wrote in July 2023. “ELEVIDYS is changing the game.”
By mid-2025, that tone had flipped. A Stocktwits user wrote after the second patient death: “I believed the hype. Now I’m looking at a 60% loss.” Another posted simply: “Trust broken.”
Analyst Commentary
Analysts initially backed Sarepta. Firms like JPMorgan and Leerink had bullish outlooks, driven by strong commercial metrics and regulatory progress. Price targets floated above $100.
After the safety disclosures, sentiment shifted. Targets dropped, and some firms halted coverage. “Regulatory momentum has stalled,” one analyst wrote in June. “It’s unclear when—or if—it will recover.”
SEC Filings & Risk Factors
In its 2024 and early 2025 filings, Sarepta acknowledged general risks of clinical failure and regulatory delay. But the filings did not mention any specific adverse events or deaths—despite evidence that the company had already been made aware.
Only after June 15 did Sarepta disclose the second death and suspend shipments. And only after June 24 did they cite liver toxicity as a known safety issue. Plaintiffs say those delays cost investors critical time to make informed decisions.
Conclusion and Investor Implications
Sarepta’s story is no longer about a promising gene therapy. It’s about what wasn’t said—and when.
Biotech stocks come with risk, but that risk must be shared honestly. Investors say they bought into a success story, not knowing the costs were rising out of frame. For others in the space, this case serves as a warning: transparency isn’t just good ethics. It’s part of the job.
Lead Plaintiff Deadline: August 25, 2025
To learn if you qualify, visit the Sarepta Therapeutics, Inc. (SRPT) Class Action Lawsuit submission page.
Attorney Advertising Disclaimer: This alert is for informational purposes only and does not constitute legal advice. Past results do not guarantee future outcomes. Consult a licensed attorney for guidance.