Introduction to Skye Bioscience, Inc. (SKYE) Securities Class Action Lawsuit
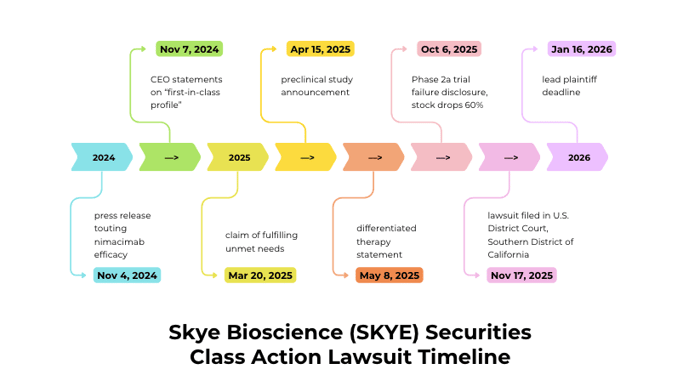
A federal securities fraud class action has been filed against Skye Bioscience, Inc. (NASDAQ: SKYE) on behalf of investors who acquired Skye securities between November 4, 2024 and October 3, 2025. Investors allege the company misrepresented the efficacy and prospects of its lead drug candidate, nimacimab, and overstated its clinical, regulatory, and commercial potential. The story turned when Skye later announced that nimacimab monotherapy failed to meet the primary endpoint in a Phase 2a trial and showed lower-than-expected drug exposure. On this news, the market sharply repriced the stock. Shareholders experienced significant losses following the corrective disclosure.
Skye Bioscience, Inc. (SKYE) Securities Lawsuit Case Details
Case Name: Stout v. Skye Bioscience, Inc., et al.
Case No.: 3:25-cv-03177-WQH-DEB
Jurisdiction: U.S. District Court, Southern District of California
Filed on: November 17, 2025
Skye Bioscience, Inc. (SKYE) Company Profile
Skye Bioscience is a clinical-stage biopharmaceutical company developing molecules that modulate G protein-coupled receptors to treat obesity, overweight, and metabolic diseases. Its lead candidate, nimacimab, is a peripherally restricted negative allosteric modulating antibody targeting the CB1 receptor involved in metabolic regulation.
Skye Bioscience, Inc. (SKYE) Securities Lawsuit Class Period
November 4, 2024-October 3, 2025, inclusive.
All persons and entities other than Defendants that purchased or otherwise acquired Skye securities during the Class Period are included in the alleged class and may be eligible to join the Skye Bioscience, Inc. (SKYE) class action lawsuit.
Allegations in the Skye Bioscience, Inc. (SKYE) Securities Class Action Lawsuit
The complaint targets Skye Bioscience, Inc. and senior executives Punit Dhillon (President and CEO), Kaitlyn Arsenault (CFO), and Christopher G. Twitty (CSO). According to investors, Defendants repeatedly described nimacimab as a differentiated, effective CB1 inhibitor positioned to meet critical needs in obesity and metabolic disease, while omitting that the drug was less effective than presented.
The narrative begins on November 4, 2024, when a company press release claimed nimacimab achieved “significant dose-dependent weight loss, fat mass reduction, lean mass preservation, and glycemic control in a [DIO] model[,]” and that preliminary data showed desired metabolic outcomes. Three days later, on November 7, 2024, CEO Punit Dhillon said the "truly peripherally-restricted CB1 inhibitor" had attributes necessary to realize benefits in the obesity landscape, and he told investors that nimacimab's "first-in-class profile" offered the right combination of efficacy and safety to drive meaningful dose and weight loss.
The optimism continued into 2025. On March 20, 2025, Dhillon asserted that nimacimab's product profile was well-positioned to fulfill “critical unmet needs.” On April 15, 2025, he said a new preclinical study showed nimacimab "effectively drives weight loss" in a DIO model. And on May 8, 2025, he stated nimacimab continued to demonstrate a differentiated profile as a potential weight loss therapy that could be set apart from GLP-1s and small-molecule CB1 inhibitors.
Investors allege that, in truth, nimacimab was less effective than represented and that its clinical, regulatory, and commercial prospects were overstated, rendering these public statements materially false and misleading.
The Truth Emerges
On October 6, 2025, Skye issued a press release with topline data from its Phase 2a CBeyond trial revealing that the nimacimab monotherapy arm did not achieve the primary endpoint of weight loss compared to placebo. The company also reported preliminary pharmacokinetic analysis showing lower-than-expected drug exposure and indicated the 200 mg weekly monotherapy dose was suboptimal. These revelations stood in stark contrast to prior assurances about nimacimab's efficacy and differentiated profile. Statements portraying nimacimab as capable of driving meaningful weight loss and meeting critical therapeutic needs were contradicted by the Phase 2a results and the exposure findings.
Market Reaction
On October 6, 2025, following the pre-market press release announcing the failed primary endpoint, Skye's stock fell $2.85 per share, or 60%, to close at $1.90 per share the same day.
Next Steps
Submissions for lead plaintiff are due: January 16, 2026.
The Court will issue its order for lead plaintiff and counsel in the weeks after submissions are due.
The Court will then consider motion for class certification.
The Court will later consider a Motion to Dismiss.
To learn if you are eligible for recovery under the SKYE securities class action lawsuit, visit the case submission page here.
![Skye Bioscience, Inc. (SKYE) Securities Class Action Lawsuit Filed [November 24, 2025]](https://dropinblog.net/cdn-cgi/image/fit=scale-down,format=auto,width=700/34260815/files/featured/skye-new-case-banner.webp)




![Perrigo Company plc (PRGO) Securities Class Action Lawsuit Filed [November 24, 2025]](https://dropinblog.net/cdn-cgi/image/fit=scale-down,format=auto,width=700/34260815/files/featured/prgo-new-case-banner.webp)
![Jayud Global Logistics Limited (JYD) Securities Class Action Lawsuit Filed [December 1, 2025]](https://dropinblog.net/cdn-cgi/image/fit=scale-down,format=auto,width=700/34260815/files/featured/jyd-new-case-banner-image.webp)
![StubHub Holdings, Inc. (STUB) Securities Class Action Lawsuit Filed [December 1, 2025]](https://dropinblog.net/cdn-cgi/image/fit=scale-down,format=auto,width=700/34260815/files/featured/stub-new-case-alert-banner-image.webp)